Titration is a method of estimation of the strength of a given substance in analytical chemistry.
These Titrations are carried out to test the strength of acids, based, buffers, redox agents, metal ions, etc.
An acid-base titration is specifically meant to estimate the strength of acids, bases, and related salts.
The principle behind the acid-base titration is neutralization. When an acid reacts with a base, it forms salt and water. This is called a neutralization reaction because theoretically pH at the endpoint is 7.
For example, when strong acid, hydrochloric acid, and strong base sodium hydroxide react, the pH at the endpoint is 7. The solution is neither acids no basic and has only water and salt.
HCl+NaOH——————>NaCl+H2O.
In an acid-base titration, a known quantity of acid is used to estimate an unknown amount of a base or vice-versa. A known reactant is taken in a burette and the test in a beaker. The reactant from the burette is added drop by drop while the beaker is swirled to enhance the reaction. This is continued until the endpoint is reached.
The reactant taken in the burette is called titrant while that taken in the flask is called as a titer

An acid-base indicator is used to indicate the endpoint of the reaction. These indicators change the color of the solution at the endpoint. In modern labs instead of indicators, pH meters are used to detect the endpoint.
5 Types of Acid Base Titration
Acid-base titration can be done in both aqueous and nonaqueous media.
A. Aqueous acid base titration
These are normal titration between acids and base dissolved in water. Hence the name aqueous titration. They are prominently used in academic labs and for standardization.
1) Strong acid V/s strong base
Here are strong acid reacts with a strong alkali to form salt and water. The reaction of this type is swift and also complete. The reaction happens in stoichiometric means, i.e., each molecule of acid reacts with the corresponding molecule of the base. At the end of the reaction, no molecule of acid or base exists as every molecule in the reaction has completely reacted to form a salt. Hence the endpoint or equivalence point is precise and sharp.
Examples of these types of acids are HCl, H2So4, HNO3, HBr, HClO4 (perchloric acid), H3PO4, etc. Examples of strong bases are NaOH, MgOH2, Al2OH3, etc.
Reaction example: HCl+NaOH—————-> NaCl + H2O
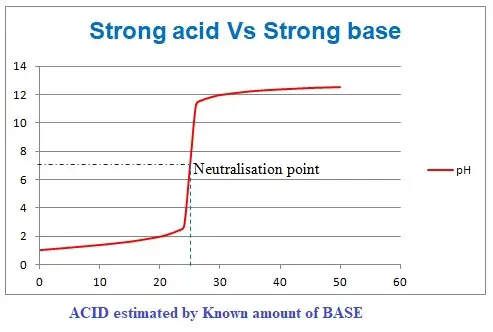
A known quantity of acid is taken in burrete to react with an unknown quantity of strong base in the flask (beaker) or vice-versa.
The pH at the endpoint is neutral, i.e., 7. So indicator changing their color around pH seven is used here.
2) Strong Acid v/s Weak Base
Here a strong base reacts with a weak acid to form salt and water. But since the reaction uses a strong acid, the pH at the endpoint will be towards acidic, i.e., below 7.
Reaction example: HCl+NH4OH—————–>NH4Cl + H2O.
Here the salt formed NH4Cl is slightly acidic. So indicators changing color at lower pH’s are employed.
During the reaction, a known concentration of strong acid is taken in a burette and allowed to react drop by drop with the base in a beaker.
3) Weak Acid V/s Strong Base
Here the reaction happens between a weak acid and a strong base. The weak acid is taken in a beaker and a known quantity of strong base is dropped from a burrete till the endpoint.
Reaction example: H2CO3+ NaOH————–>Na2Co3+H2O

The salt formed is slightly basic, so the pH at the endpoint is above 7. The indicator used is one with a change in color at higher pHs.
4) Weak Acid V/s Weak Base
Here both acid and base are weak. So mostly they are avoided due to imprecise endpoints. At the endpoint, the pH will be seven theoretically. But cannot be measured precisely like that in strong acid and strong base case. An extra amount of titrant is needed to reach the endpoint due to the imprecise reaction.
Reaction example: H2CO3+NH4OH————–>NH4OH+H2O
The endpoint is neutral as the salt is neutral, but due t excess titrant added the pH could be in favor of it.
Check the video below for method
Back titration or Indirect titration
Even the substance is not acidic or basic it can still be estimated. For this, the substance is converted by the use of some reaction and then estimated employing a back titration method.
Example: Estimation of aspirin. Aspirin is a weak acid drug. So to the sample of aspirin in a beaker, a known volume sodium hydroxide is added. It is allowed to react by heating. Once the reaction is over, the sodium remained unconsumed is estimated by back titration with concentrated hydrochloric acid
Step. 1) Aspirin + NaOH (Excess) —————> Alkaline hydrolysis + Remained NaOH.
Step. 2) Remained NaOH + HCl —————-> NaCl + H20.
B. Non-Aqueous titration:
These are conventional methods of non-aqueous titration. Here instead of water as solvent glacial acetic acid is used to make the reactants. They are similar to the above types of acid-base reactions. Since many drugs are water-insoluble and slightly acidic or basic, they are analyzed by non-aqueous titrations. They are extensively used for quality control and analysis of drugs.
Reaction example: psuedoephidrin+HCl

This material was easily followed by certificate standard class. Thanks
Thanks.. This material is very good