Chemical reaction is the way by which the life functions on the earth.
Most changes in the matter takes place by some sort of chemical reaction.
The whole science of chemistry revolves around chemical reactions.
There are many types of chemical reactions to suit different needs of the matter.
In general a chemical reaction brings a significant change to the parent molecule involved.
Almost all the chemical reaction happen in liquid form or gaseous form.
So for the chemical reaction to happen, the molecules must be freely moving.
Chemical reaction are many and they are studied in organic chemistry, inorganic chemistry, biochemistry, phyto-chemistry etc.
In general chemical reactions are used for purposes like synthesis of new molecule, analysis of a test substance, identification of a substance, degradation or break down of a molecule, etc.
Types of Chemical Reactions
1. Neutralization reactions: These are the reactions which occur in between and acid and base. These reactions result in formation of water and salt in general. The end product can also be a weak acid or base if weak base or weak acid is used in the reaction.

The pH at end point is 7 for a reaction between strong acid and strong base. But it can above 7 for reaction between strong base and weak acid. And below pH7 if the reaction is between strong acid and weak base. These reactions are used in industry and research for analysis of compounds by acid-base titrations. Also this reaction is used to treat stomach acidity in health care by use of antacids.
2. Redox reactions: These reactions are also called as oxidation-reduction reactions. The redox reactions mostly involve oxidation of one substance while other substance is reduced. Oxidation involves either
- Either addition of oxygen
- Removal of hydrogen
- Or removal of electrons
Reduction involves either
- Removal of oxygen.
- Addition of hydrogen.
- Addition of electron.
3. Complexation reaction: This is a reaction which leads to formation of an insoluble chemical complex. These reactions are done for sake of analysis, quality control or to treat poisoning.
4. Substitution reactions: These are the reactions in which an atom, element or chemical group is removed from the previous molecule. This type of reaction is also termed under displacement reaction.
5. Addition reactions: These are the reactions in which there is chemical addition. I.e. there is an addition of chemical group, element or an atom to the main molecule in the reaction.

This can be a synthesis reaction in making of new compounds.
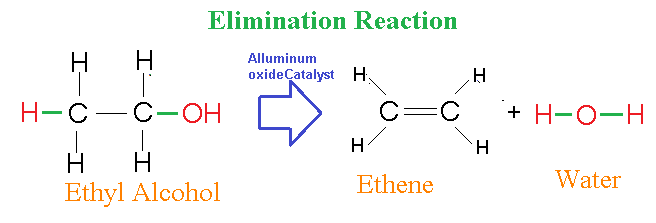
6. Elimination reactions: As the name indicates, these are the reactions in which there is elimination of an atom, chemical group or element from the main molecule.
Below is an example of a reaction where from Ethanol molecule, water is removed off to form ethylene or ethene.
7. Rearrangement reactions: These are a different set of reactions where is there is neither addition or nor elimination of an atom, chemical group or an element from the parent molecule. Here the parent molecule is just changed in terms of chemical structure and configuration. Many isomers come under this type of reactions.
