Different types of titrations are used in the field of analytical chemistry to evaluate chemical compounds like the
- Acid-base titrations
- Redox titrations
- Precipitation titrations
- Complexometric titrations.
But, based on the experimental requirement and conditions, there are few more types as the nonaqueous, iodometric, indirect titrations, etc.
We will see all the methods in detail.
Definition of titration
It is a method of analysis wherein a test substance is allowed to react with a known standard solution in the presence of an indicator until the endpoint.
An endpoint is one where the test substance has been completely reacted with the analyzing regent.

The reagent added from burrete to estimate is called a titrant. The substance underestimation is called titrand.
The different types of titration are
A) Based on the method of titration
They are of three types of titration based on the method used in the process of titration.
1. Direct titration
- As the name indicates, it is a conventional titration.
- A known amount of titrant is added from a burrete to a titrand sample taken in a flask.
- Here one substance is analyzed for its quantity by another substance of known volume and concentration.
2. In-direct titration
- Theoretically, it is converting a substance into acid and analyzing it with a base. (also vice-versa).
- This is a method extrapolated to use titration for non readily reactive substances.
- A substance can be weakly acidic and so it does not permit a precise analysis by direct titration.
- So first that substance is chemically altered to be more reactive in acidic or basic form and then analyzed by adding a titrant.
3. Back titration
- This method is also suitable for weakly reactive or non-reactive substance estimation.
- Here a substance is allowed to react with excess and known quantity of a base or an acid.
- The remnant excess base or acid is estimated by a known quantity of acid or base deceptively.
It is called back titration as we are estimating a substance that was added by us.
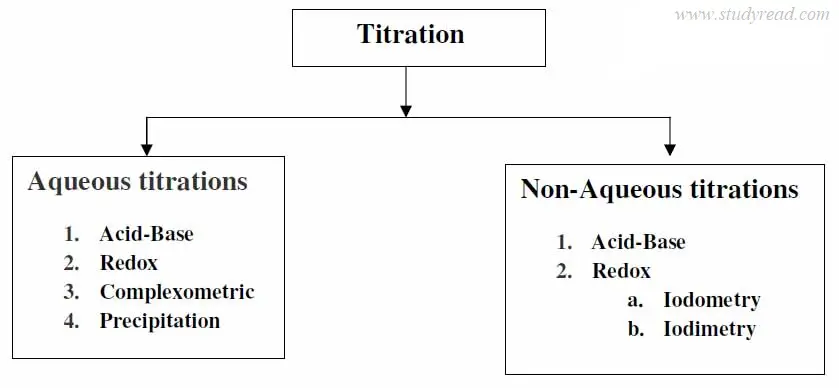
B) Based on the nature of solvents and chemical reaction
The titrations can also be classified based on the nature of solvents used.
Based on the solvent used, they can be classified as aqueous and non-aqueous types.
Further, based on the nature of the chemical reaction, they are classified as below.
Aqueous titrations
1. Acid-base titrations. ( neutralization):
- An unknown sample of acid is estimated with a known quantity base or vice-versa.
- The result reaches a neutral point at pH-7 and in most cases, salt is formed.
Check out Types of acid-base titration for more.
2. Redox titrations
- It is also called oxidation-reduction titration.
- Here a reducing agent is allowed to react with an oxidizing agent till the endpoint.
- The common oxidizing agents used are Potassium permanganate, bromine, cerium, etc.
3. Complexometric titrations
- As the name indicates, the endpoint is seen by the formation of a complex molecule.
- Here titrant and titrand react to form a complex till the endpoint is reached.
- Once complex is formed, the complex is stable and no further reaction takes place.
- The reaction depends on the chelating agent’s ability to form a complex with the sample under test.
An example of chelate is ethylene tetra-acetic acid (EDTA)sodium salt.
4. Precipitation titrations
- The reaction occurs by the formation of a solid precipitate at the bottom of the flask.
- Here titrant reacts with titrand to form an insoluble precipitate.
An example of such a reaction is Silver nitrate with Ammonium chloride.
- The reaction forms a white precipitate of silver chloride.
2. Non-Aqueous titrations
These are the titration done for organic and medicinal compounds.
So widely used in medicinal chemistry and pharmaceutical labs.
a) Acid-base titrations. (neutralization):
Here the reaction occur in organic solvents like glacial acetic acid, and the acid used is perchloric acid (HClO4)
b) Redox titrations.
Here the reaction is done as iodometry or iodimetry. Iodine is used as an oxidizing agent, and a reducing agent is thiosulfate.
c) Iodometry
Here the sample is made to release iodine from within, and this released iodine is measure with sodium thiosulfate as a reducing agent.
d) Iodimetry
Here the sample under test is measured with a known concentration of Iodine.
These are similar to aqueous titrations, but instead of water as a solvent, an organic solvent is used here.
C) Based on the strength of reagents used
The chemical reaction involved in titration is the main principle of titration. But the chemical reactions also depend on the nature and strength of the reagents used in the reaction.
So acid-base titrations or the neutralization reactions are also further divided as
- Strong acid v/s Strong Base: Here both acid and base are strong. Ex: HCl v/s NaOH. Here the endpoint would be at an exact pH of 7. The salt formed in reaction would be neutral.
- Strong acid v/s Weak Base: A strong acid reacts with a weak base. Ex: HCL v/s NH4OH. Here the endpoint would go towards acidic pH, i.e., below 7. The salt formed in the end would be slightly acidic.
- Weak acid v/s Strong Base: A weak acid reacts with a strong base. Ex: Acetic acid v/s NaOH. Here the endpoint would be tilting towards basic pH, i.e., above 7. The salt formed during neutralization would be slightly alkaline.
- Weak acid v/s Weak Base: Reaction between a weak acid and a weak base. Ex: Acetic acid v/s NH4OH. The endpoint would have a pH of 7. Here the salt formed as a final product would be neutral.
A titrand is taken in a beaker while the titrant is dropped from a burette. The sole purpose of titration is for qualitative and quantitative estimation of a given substance.
These methods are comparatively economic and easy to perform. Further, the technique is also reproducible across all the labs. Hence, they are routinely used in industry, research, and also education.
Calculation: All the processes of titration are noted in a chemistry lab report manually. This includes the molar concentration of solutions, their volumes, indicators, formulas, etc. This helps to calculate final values.



Hi Catalin! Sorry to mention but we do not have methods for crafting emerald armour.
This is cool and all, but do you guys know a method for crafting emerald Armour?
Good
Nice information
Nice
I want as a pdf.
Very Nice information.
Nice gathering of information at a time
you are making me more confuse with this type of answers. be more straight and specific next time
Kanishk! seems you are talking about acid value of oils and fats.
It’s because it takes more KOH per liter of oil to achveie the same reaction. It has to do with KOH’s molar chemical ratio vs. NaOH’s. ie. NaOH is more potent, so it takes less per liter of oil to cause a successful reaction.