You might have gone through different types of distillation techniques. Of them, azeotropic distillation is quite an interesting one.
This method is used to separate azeotropic solutions. As per Wikipedia, an azeotrope is a mixture that cannot be separated by simple distillation.
Most liquid mixtures can be separated by fractional distillation (FD). During the fractional distillation, one of the components gets into the vapor phase. This vapor is separated, cooled, and collected in a receiver. The other components of the mixture remain.
But this does not happen with azeotropic solutions. When these solutions are subjected to FD, both mixtures are in the vapor form. Hence, they cannot be separated. This is due to similar vapor pressures of liquids. The azeotropic distillation technique is employed to solve this problem.
Here, a third substance is added to an azeotropic solution. This addition changes the relative volatility of the components.
For example, glycerin is added to a mixture of water and alcohol (rectified spirit).
The addition of glycerin forms a new azeotrope between water and glycerin. This is because both are more polar than ethyl alcohol.
The result is that the volatility of ethyl alcohol is enhanced. Hence, it can be distilled separately, leaving water and glycerin behind.
Instead, if you add benzene to the above mixture, it forms a new azeotrope with ethyl alcohol. So, water can be removed from the mixture. Later, even the azeotrope breaks, and you can remove benzene to get absolute alcohol.
This distillation requires a simple distillation apparatus. Hence, the method is less expensive and also faster.
Procedure of distillation
1. Take 100ml of rectified spirit and determine the concentration of water in it. Also, note down its refractive index
2. Add 5 ml of glycerine to the above solution and transfer the entire mixture to a round bottom flask.
3. As shown below, Assemble the flask into the simple distillation apparatus.
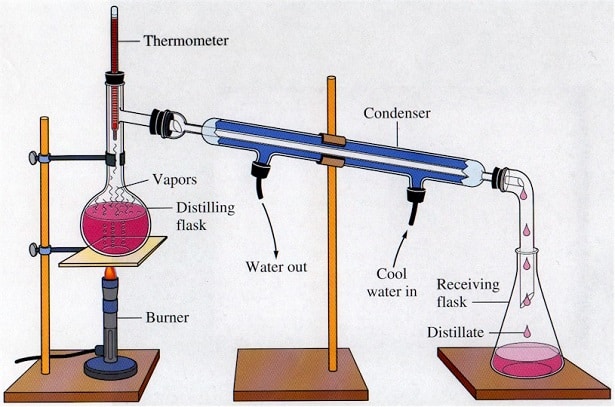
4. Heat the mixture and note the temperature of the alcohol distillation.
5. The alcohol vapors rise into the condenser and then cool separately into the receiver flask.
6. Note down the refractive index of absolute alcohol to determine the percentage.
Applications of azeotropic distillation
Azeotropic distillation is mainly used for
1. Preparation of Absolute alcohol. Alcohol is available in different concentrations, like 70% and 90%, in combination with water. Absolute alcohol is 100% alcohol without any parts of water.
2. This distillation is also used in petroleum refineries.
