The pharmacological management of Type 2 Diabetes Mellitus (T2DM) has undergone a major shift over the last decade, moving from a “glucose-centric” approach to one focused on organ protection.
As we navigate the clinical landscape of 2026, two oral agents remain at the forefront of therapy: Januvia (Sitagliptin) and Jardiance (Empagliflozin).
While both drugs effectively lower HbA1c (blood glucose levels), they achieve this through completely different physiological mechanisms.
Januvia, a DPP-4 inhibitor, functions by enhancing the body’s internal hormonal response to meals.
In contrast, Jardiance, an SGLT2 inhibitor, bypasses insulin-dependent pathways entirely and relies on the renal system to eliminate excess glucose from the body.
Januvia vs Jardiance Comparison Table
| Feature | Januvia (Sitagliptin) | Jardiance (Empagliflozin) |
|---|---|---|
| Primary Indication | Type 2 Diabetes (T2DM) | T2DM, Heart Failure, CKD |
| A1c Reduction | 0.5% – 0.8% | 0.7% – 1.0% |
| Weight Impact | Weight Neutral | Weight Loss (2–3 kg avg) |
| Cardiovascular (CV) Outcome | Neutral (Safe) | Protective & Superior (Reduced CV death by ~38%) |
| Renal (Kidney) Impact | Requires dose reduction in CKD | Nephroprotective (Slow CKD progression) |
| 2026 Cost Status | Low-cost Generic | Brand Name (Premium) |
This article is for educational purposes only and does not constitute medical advice. Always consult a healthcare professional before starting any treatment.
Table of Contents
The 2026 Clinical Context
This year marks a pivotal moment for healthcare providers and students alike.
With major patent milestones for Januvia occurring throughout 2026, the arrival of more affordable generic Sitagliptin is lowering the “entry barrier” for access to Incretin-based therapies.
Simultaneously, Jardiance has moved beyond its identity as only a “diabetes drug” to securing its position as a foundational treatment for Heart Failure (HF) and Chronic Kidney Disease (CKD)—regardless of a patient’s diabetic status.
Understanding the pharmacological nuances between these two agents is no longer just about blood sugar alone.
It is about matching the drug’s molecular mechanism to the patient’s long-term cardiovascular and renal prognosis.
II. Pharmacological Mechanism of Action
Both medications are oral antihyperglycemic agents, but they belong to different pharmacological classes and modulate glucose levels through entirely different physiological pathways.
1. Januvia (Sitagliptin): The Incretin Enhancer
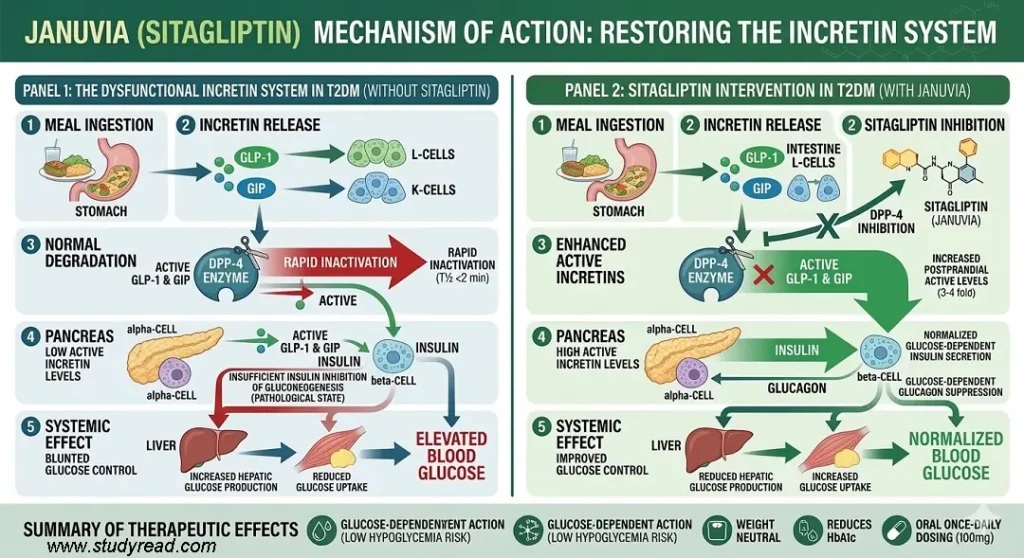
Januvia acts as a Dipeptidyl Peptidase-4 (DPP-4) inhibitor. It works by protecting the body’s natural “Incretin” hormones (GLP-1 and GIP), which are usually degraded within minutes by the DPP-4 enzyme.
- Target: DPP-4 Enzyme.
- Mechanism: By blocking this enzyme, Januvia increases the levels of active GLP-1 and GIP.
- Result: These hormones GLP-1 and GIP, signal the pancreas to increase insulin and decrease glucagon only when blood sugar is high.
Key Advantage: Because this process is glucose-dependent, Januvia has a very low risk of causing hypoglycemia when used as a monotherapy.
2. Jardiance (Empagliflozin): The Renal Glucose Shunt
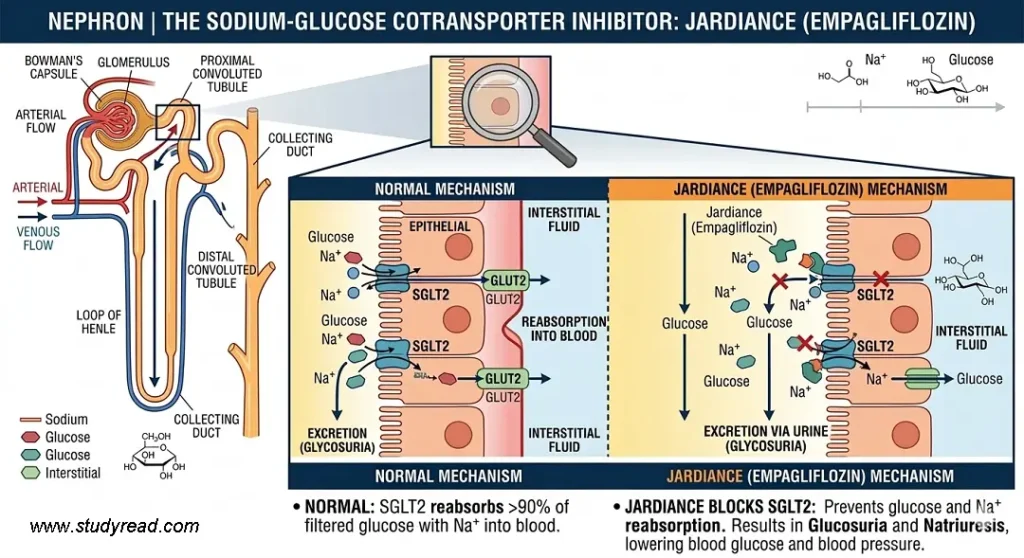
Jardiance belongs to the SGLT2 inhibitor class. Unlike Januvia, its primary site of action is the kidneys rather than the pancreas.
- Target: Sodium-Glucose Cotransporter 2 (SGLT2) in the Nephron.
- Mechanism: In the nephron, it blocks the protein responsible for reabsorbing glucose from the urine back into the blood.
- Result: Excess glucose is “shunted” out of the body through urine (Glycosuria), which also leads to a reduction in blood pressure and weight.
So, unlike Januvia, Jardiance works independently of insulin and the pancreas.
- Normally, the kidneys filter and reabsorb approximately 180g of glucose daily to maintain energy. SGLT2 is responsible for 90% of this reabsorption.
- Jardiance inhibits this transporter in the Proximal Convoluted Tubule (PCT) of the nephron in the kidney.
- Instead of being pulled back into the bloodstream, excess glucose and sodium are excreted through the urine.
- Caloric Loss: Excreting glucose results in a ~200–300-calorie loss per day, which drives the weight-loss benefit.
- Osmotic Diuresis: The movement of salt with glucose leads to a mild diuretic effect, reducing blood pressure and easing the workload on the heart.
Comparison Table: Molecular Impact
| Feature | Januvia (DPP-4i) | Jardiance (SGLT2i) |
|---|---|---|
| Primary Organ | Pancreas / Liver | Kidneys |
| Pathway | Insulin-Dependent (Incretin) | Insulin-Independent |
| Effect on Weight | Neutral | Reduction (Caloric loss) |
| Effect on BP | Neutral | Reduction (Natriuresis) |
| Main Molecular Goal | Increase GLP-1/GIP levels | Decrease glucose reabsorption |
Case Study: Choosing Between Cardioprotection and Neutrality
In clinical practice, the choice between Januvia and Jardiance often comes down to the patient’s cardiovascular history rather than just HbA1c levels.
Consider a 64-year-old female patient with Type 2 Diabetes and stage 2 Chronic Kidney Disease (eGFR 55 mL/min).
While Januvia (a DPP-4 inhibitor) is weight-neutral and excellently tolerated for glucose control, it offers no direct protection for her heart or kidneys.
In this case, the transition to Jardiance (an SGLT2 inhibitor) was preferred. Why? Because the latest 2025/2026 data—including the EMPACT-2025 findings—confirms that Jardiance provides a 35% reduction in heart failure hospitalizations.
For this patient, we weren’t just treating a blood sugar number; we were protecting her heart from future failure.”
III. Why 2026 is the “Tipping Point.”
The most important takeaway for 2026 is Accessibility vs. Benefit:
- The Budget Option: With the launch of generic Sitagliptin in Mid–2026 (by manufacturers like Sandoz and Apotex), Januvia has become the most affordable “add-on” to Metformin.
- The Premium Option: Jardiance remains the “gold standard” for patients with comorbidities such as Heart Failure or Chronic Kidney Disease, as it is now FDA-approved for these conditions, even in patients without diabetes.
IV. Key Clinical Benefits & 2026 Updates
The clinical choice between Januvia and Jardiance is dictated by two primary factors: Organ Protection vs. Patient Tolerability.
1. The “Jardiance Advantage”: Beyond Glycemic Control
Jardiance (Empagliflozin) has transitioned from a second-line diabetes agent to a foundational therapy for cardio-renal health.
Expanded FDA Indications (2026 Status):
- Heart Failure (HF): Jardiance is now universally indicated for heart failure across the entire spectrum of Left Ventricular Ejection Fraction (LVEF).
- Whether a patient has HFrEF (reduced) or HFpEF (preserved), Jardiance significantly reduces the risk of CV death and hospitalization.
- Chronic Kidney Disease (CKD): Following the landmark EMPA-KIDNEY trials, Jardiance is officially indicated in 2026 to slow the progression of CKD in adults, even those without Type 2 Diabetes.
- It reduces the risk of sustained eGFR decline and end-stage kidney disease (ESKD).
- Metabolic “Bonus”: Unlike most oral agents, Jardiance promotes a caloric deficit through glycosuria.
- This can result in an average weight loss of 2–3 kg and a modest reduction in systolic blood pressure.
2. The “Januvia Advantage”: Reliability and Accessibility
While Jardiance dominates in “protection,” Januvia (Sitagliptin) wins in tolerability and affordability (as of 2026).
Superior Tolerability Profile:
- GU Infection Avoidance: The most significant “Januvia Advantage” is the absence of genitourinary risks.
- For patients prone to recurrent Urinary tract infections (UTIs) or yeast infections (a common side effect of SGLT2 inhibitors like Jardiance), Januvia is the safer alternative.
- Weight Neutrality: For patients who are already at their optimal body weight or at risk of lower body weight (e.g., the elderly), Januvia’s weight-neutral profile is often preferred over the weight loss associated with Jardiance.
- Cost-Effectiveness: With earlier patent settlements allowing generic entry in mid-2026, Sitagliptin has become the most budget-friendly “add-on” to Metformin.
- Medicare Impact: Following the 2026 Medicare price negotiations, the brand-name Januvia cost has dropped to $113/month, making it more accessible to a much wider patient demographic in the USA than in previous years.
| Patient Profile | Preferred Choice | Rationale |
|---|---|---|
| History of Heart Failure | Jardiance | Superior CV mortality reduction. |
| Recurrent UTIs | Januvia | Avoids glycosuria-induced infections. |
| CKD (eGFR 30-60) | Jardiance | Nephroprotective (slows progression). |
| Strict Financial Budget | Januvia | Availability of low-cost generics (2026). |
| Need for Weight Loss | Jardiance | Promotes 4–6 lbs of loss on average. |
V. Side Effects and Safety Profile
While both Januvia and Jardiance are generally well-tolerated, their side-effect profiles stem from their distinct pharmacological mechanisms.
1. Januvia (Sitagliptin): The Low-Impact Option
Because Januvia works by enhancing the body’s natural incretin system, its side effects are typically mild and systemic.
- Common Reactions: The most frequent reports involve nasopharyngitis (stuffy/runny nose, sore throat) and upper respiratory tract infections.
- Severe Joint pain (Arthralgia): In some patients, DPP-4 inhibitors have been linked to severe and disabling joint pain. This can occur anywhere from one day to years after starting the medication, but typically resolves on discontinuation.
- Pancreatitis Warning: Though clinical trials (like TECOS) showed no increased risk, post-marketing reports have identified cases of acute pancreatitis. Patients should be counseled to report persistent, severe abdominal pain immediately.
- Bullous Pemphigoid: A rare autoimmune skin condition characterized by large, fluid-filled blisters. If suspected, the drug must be stopped immediately.
2. Jardiance (Empagliflozin): The Renal-Driven Profile
Jardiance’s side effects are almost exclusively “mechanistic”—meaning they are a direct result of moving sugar and water through the kidneys.
- Genitourinary Infections (GU): This is the most common side effect. Because Jardiance causes glycosuria (sugar in the urine), it creates a breeding ground for bacteria and fungi.
Note: Female yeast infections are more common, but male genital infections (balanitis) also occur.
- Volume Depletion & Hypotension: By acting as a mild osmotic diuretic, Jardiance can cause a drop in blood pressure. This leads to dizziness, lightheadedness, or fainting, particularly in elderly patients or those already taking “water pills” (diuretics).
- Euglycemic Ketoacidosis (DKA): This is a rare but life-threatening emergency where the blood becomes too acidic, but blood sugar levels remain near normal (under 250 mg/dL). This “masked” high sugar makes it harder to diagnose.
- Fournier’s Gangrene: An extremely rare but severe bacterial infection of the tissue under the skin in the perineal area. Any redness or tenderness in the genital region accompanied by fever requires urgent surgical evaluation.
| Side Effect | Januvia | Jardiance |
|---|---|---|
| UTI / Yeast Infection | Low (Same as placebo) | High (Due to glycosuria) |
| Blood Pressure | Neutral | May Decrease (Beneficial for some) |
| Pancreatitis Risk | Rare (Monitor closely) | Minimal |
| Ketoacidosis Risk | Minimal | Rare (Euglycemic DKA) |
| Joint Pain | Possible (Severe Arthralgia) | Minimal |
VI. Dosing and Administration
Both medications offer the convenience of once-daily oral dosing, typically taken in the morning.
However, their renal adjustment protocols differ significantly due to their unique mechanisms of action.
1. Januvia (Sitagliptin): The “Dose-Reduction” Model
Since Januvia is primarily excreted by the kidneys, the dose must be adjusted to prevent drug accumulation and potential toxicity as kidney function declines. The goal is to maintain a consistent therapeutic plasma concentration.
- Standard Dose: 100 mg once daily.
Renal Adjustments (Based on eGFR):
- eGFR ≥ 45: No adjustment needed (100 mg).
- eGFR 30 to < 45: Reduce dose to 50 mg once daily.
- eGFR < 30 (including Dialysis/ESRD): Reduce dose to 25 mg once daily.
Clinical Note: Januvia can be taken at any time of day, regardless of the timing of dialysis.
2. Jardiance (Empagliflozin): The “Indication-Specific” Model
Jardiance dosing in 2026 is unique because the “cutoff” for stopping the drug depends on why the patient is taking it.
- Standard Dose: 10 mg once daily. Can be increased to 25 mg if additional glycemic control is needed and the patient has an eGFR ≥ 45.
Renal Considerations:
- For Glycemic Control (T2DM): Not recommended if eGFR < 30. Because its mechanism relies on filtering glucose through the nephron, it simply becomes ineffective at lowering blood sugar once the filtration rate is too low.
- For Heart Failure (HF) & CKD: Can be initiated in patients with an eGFR as low as 20 (for HF) or 25 (for CKD).
- Continuation Rule: In 2026, guidelines suggest that if a patient is already on Jardiance for heart or kidney protection, it may be continued even if the eGFR drops below 20, up until the point they initiate dialysis.
Surgical Hold: Must be withheld for at least 3 days before major surgery to reduce the risk of euglycemic ketoacidosis.
👉 The choice between Januvia and Jardiance is no longer just about blood sugar; it is about choosing the right “side effect” and the right “protective” profile.
VII. Conclusion: The 2026 Clinical Verdict
The choice between Januvia and Jardiance is no longer just about blood sugar; it is about choosing the right “side effect” and the right “protective” profile.
1. Choose Januvia (Sitagliptin) if:
- Budget is the Primary Concern: With the May 2026 generic rollout, Sitagliptin has become the most affordable add-on to Metformin.
- It is the gold standard for patients who lack premium insurance coverage or are affected by the 2026 “patent cliff.”
- High Sensitivity to Infections: For patients with a history of recurrent UTIs or yeast infections, Januvia is the safer choice as it does not promote glycosuria.
- Need for Stability: Choose Januvia if the patient is already at their ideal weight and does not require a medication that interferes with their current body composition or blood pressure.
- Elderly or Frail Patients: Its once-daily dosing at any time of day and lower risk of dehydration make it a “set-and-forget” medication for geriatric care.
2. Choose Jardiance (Empagliflozin) if:
- Organ Protection is Required: If the patient has established Heart Failure (HFrEF or HFpEF) or Chronic Kidney Disease (CKD), Jardiance is the mandatory choice due to its proven ability to reduce CV death and slow renal decline.
- Weight Loss is preferred: For patients with high BMI, the modest weight loss (avg. 2–3 kg) achieved with SGLT2 inhibition is a significant clinical advantage.
- Blood Pressure Management: Jardiance provides a “free” reduction in systolic blood pressure (approx. 3–5 mmHg), which can help simplify a patient’s overall antihypertensive regimen.
- Pediatric Cases: Jardiance is FDA-approved for children ages 10 and older, whereas Januvia remains an adult-only therapy in 2026.
Whereas Januvia remains an adult-only therapy after failing to meet efficacy endpoints in pediatric trials
Summary Table: Final Recommendation
| Feature | Januvia | Jardiance |
| Best For | Cost & Tolerability | Heart & Kidney Protection |
| Weight Impact | Neutral | Loss |
| Infection Risk | Low | Moderate (UTIs) |
| 2026 Price | Low (Generic) | High (Brand) |
| First-Line “Add-on”? | If cost is a barrier | If CV risk is high |
| Medication | 2023 List Price | 2026 Negotiated Price | % Discount |
| Januvia (Sitagliptin) | $527 | $113 | 79% |
| Jardiance (Empagliflozin) | $573 | $197 | 66% |
References:
- FDA approves empagliflozin for heart failure regardless of ejection fraction
- Sodium-Glucose Transport 2 (SGLT2) Inhibitors
- Dipeptidyl Peptidase IV (DPP IV) Inhibitors
- Empagliflozin in Patients with Chronic Kidney Disease
- Medicare Drug Price Negotiation Program: Negotiated Prices for Initial Price Applicability Year 2026. U.S. Department of Health and Human Services.
- Pancreatic Safety of Sitagliptin in the TECOS Study
- MON-522 Sitagliptin-Induced Bullous Pemphigoid: A Rare But Devious Side Effect.
- A Case of Severe Metabolic Acidosis due to Jardiance-Induced Euglycemic Diabetic Ketoacidosis.
- FDA warns about rare occurrences of necrotizing fasciitis or Fournier’s gangrene
