For decades, the American medical approach to obesity and Type 2 Diabetes was rooted in a “lifestyle-first” model that often failed to account for complex biological resistance.
As we move into 2026, we are witnessing a fundamental paradigm shift.
Obesity is no longer viewed through the lens of willpower, but as a chronic, relapsing metabolic disease driven by hormonal dysregulation.
At the center of this revolution are GLP-1 (Glucagon-Like Peptide-1) receptor agonists.
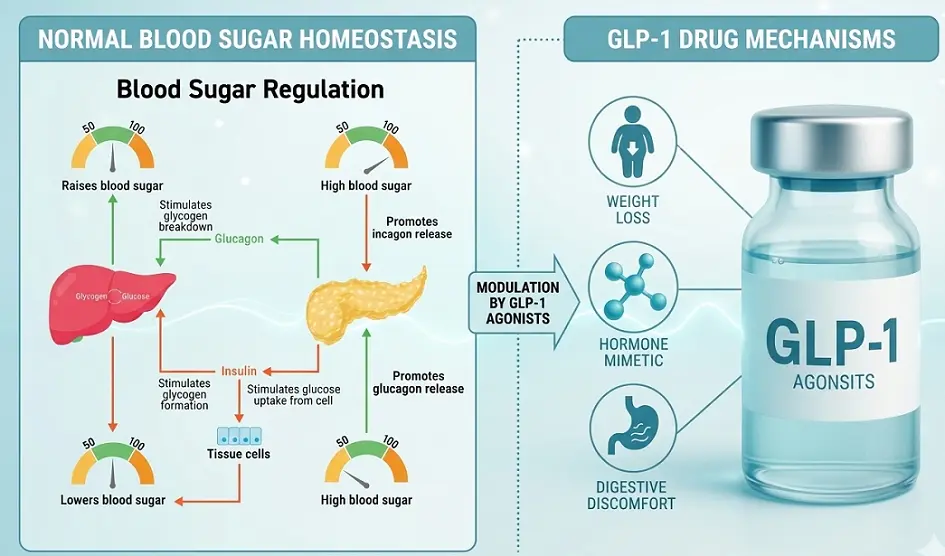
Originally developed for glycemic control in diabetics, these molecules have transcended their original purpose.
In the last 12 months, the FDA has expanded its indications to include everything from cardiovascular protection and chronic kidney disease (CKD) to obstructive sleep apnea (OSA) and liver fibrosis (MASH).
However, the rapid expansion of this drug class has created a confusing landscape of brand names and dosages.
While the media often uses “Ozempic” as a catch-all term, the pharmacological reality is much more nuanced:
- Ozempic and Wegovy share the same active molecule (Semaglutide), but differ significantly in their FDA-approved indications and clinical strengths.
- Zepbound represents the next generation of “multi-agonists,” utilizing Tirzepatide to target two separate hormonal pathways simultaneously.
- The 2026 “Pill” Launch: With the January 2026 launch of Oral Wegovy (25 mg), the era of Injectable therapy is giving way to more accessible, needle-free options.
This article provides a side-by-side pharmacological breakdown to help healthcare students, providers, and patients understand which “pen” (or pill) is truly the most effective for specific metabolic goals.
GLP 1 agonist comparison chart
| Feature | Ozempic | Wegovy | Zepbound |
| Active Molecule | Semaglutide | Semaglutide | Tirzepatide |
| Drug Class | Selective GLP-1 RA | Selective GLP-1 RA | Dual GIP & GLP-1 Agonist |
| FDA Status (2026) | T2 Diabetes / MACE | Obesity / MACE / MASH | Obesity / Obstructive Sleep Apnea |
| Max Weekly Dose | 2.0 mg | 2.4 mg (or $7.2 mg*) | 15.0 mg |
| Weight Loss (Avg) | 6% – 10% | 14.9% | 20.9% |
| CV Benefit (MACE) | Proven 26% Reduction | Proven 20% Reduction | Non-inferior (Studies Ongoing) |
| Primary Mfr. | Novo Nordisk | Novo Nordisk | Eli Lilly |
*Note: As of early 2026, higher doses of semaglutide (7.2 mg) and oral Wegovy (25 mg) have entered the US market to compete with Tirzepatide’s potency.
2. Mechanism of Action of GLP-1 agonists
The efficacy of GLP-1 agonists stems from three specific physiological interventions that rewire the body’s energy handling.
I. The Incretin Effect (Pancreatic Insulin Stimulation)
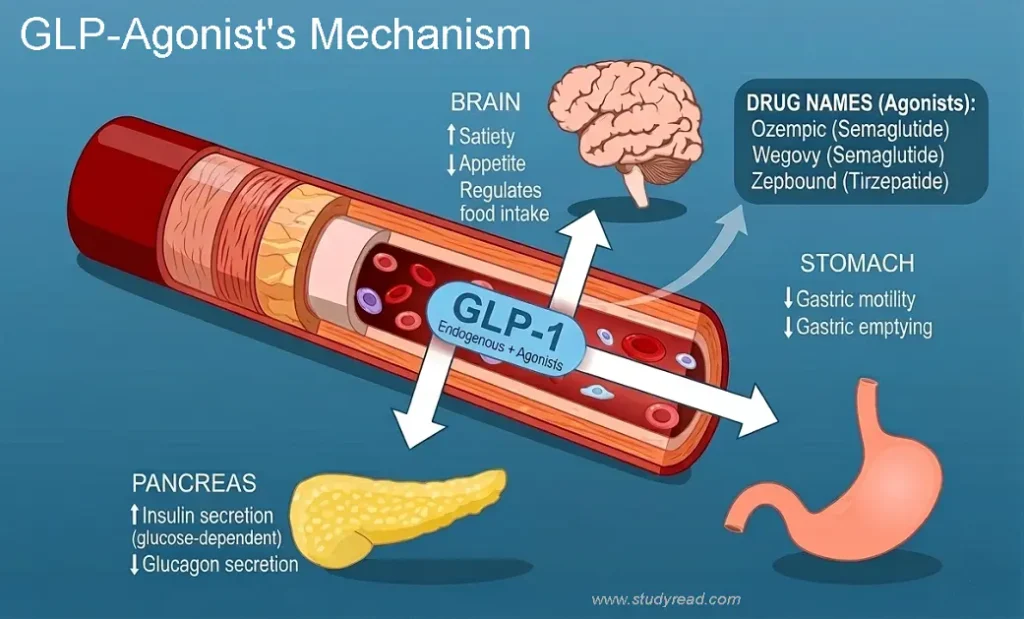
These drugs are incretin mimetics.
They mimic the natural GLP-1 hormone released by the L-cells in the intestine.
- The Action: They stimulate the pancreas to release insulin, but only when blood sugar is high.
- Weight Loss Impact: By stabilizing insulin and preventing “blood sugar spikes,” they reduce the metabolic drive to store fat.
II. Inhibition of Glucagon (Liver Glucose Control)
Glucagon is the “opposite” of insulin; it tells the liver to dump sugar into the blood.
- The Action: GLP-1 agonists inhibit the secretion of glucagon from pancreatic alpha cells.
- Weight Loss Impact: When glucagon is low, the body stops producing “new” sugar and begins to favor burning stored glycogen and fat for energy.
III. Gastric Emptying & Satiety (Stomach Fullness)
This is the mechanism most responsible for the “fullness” sensation.
- The Action: GLP-1 receptors in the stomach slow down gastric motility.
- Food stays in the stomach longer, physically signaling the vagus nerve that you are full.
- The Brain Connection: Beyond the stomach, these molecules cross the blood-brain barrier to target the hypothalamus, effectively “muting” the food-noise and cravings that lead to overeating.
| Medication | Molecule | Form | 2026 Primary Indication | Avg. Weight Loss |
| Zepbound | Tirzepatide | Injection | Obesity / Sleep Apnea (OSA) | 20.9% |
| Wegovy (Shot) | Semaglutide | Injection | Obesity / Heart Health (MACE) | 14.9% |
| Wegovy (Pill) | Semaglutide | Daily Tablet | Chronic Weight Management | 16.6% |
| Ozempic | Semaglutide | Injection | Type 2 Diabetes / Kidney (CKD) | 6% – 9% |
| Rybelsus | Semaglutide | Daily Tablet | Type 2 Diabetes | 4% – 5% |
3. Comparison of the “Big Three” (Injectables)
While all three are administered via once-weekly subcutaneous injection, their pharmacological profiles and FDA-approved “extra” benefits differ significantly as of 2026.
I. Ozempic (Semaglutide)
- Primary Role: Type 2 Diabetes (T2D) management.
- The 2026 Edge: Ozempic remains the “gold standard” for Cardiovascular-Kidney-Metabolic (CKM) syndrome.
- Key Clinical Data: The FLOW trial confirmed that Ozempic reduces the risk of major kidney disease events by 24% in T2D patients.
- It is the preferred choice for patients where renal protection is as important as glycemic control.
- Average Weight Loss: Approximately 6–7% (at T2D dosages).
While GLP-1 agonists like Ozempic directly mimic the hormone, some patients may first be prescribed DPP-4 inhibitors like Januvia to prevent the breakdown of the body’s natural incretins.
II. Wegovy (Semaglutide)
- Primary Role: Chronic Weight Management (Obesity).
- The 2026 Edge: Beyond weight loss, Wegovy is now a primary tool for Preventive Cardiology.
- Key Clinical Data: The SELECT trial proved that Wegovy reduces the risk of major adverse cardiovascular events (MACE)—like heart attack and stroke—by 20% in adults with heart disease and obesity, even those without diabetes.
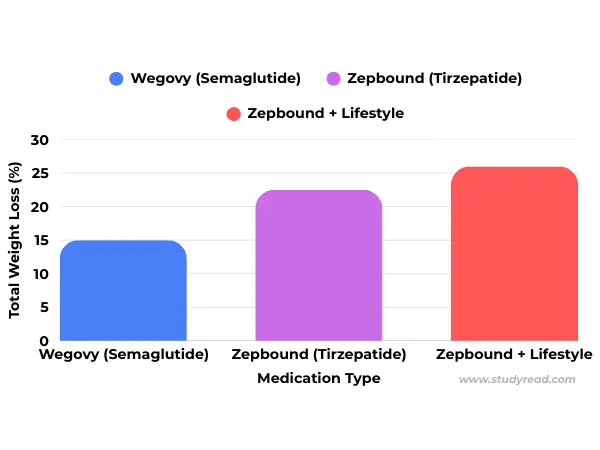
- Average Weight Loss: Approximately 14.9%
Remember, Wegovy Pill is essentially a high-dose version of Rybelsus, specifically rebranded and titrated for obesity. In 2026, many US insurance companies will only cover the 25 mg version under an “Obesity” diagnosis, while Rybelsus stays under “Diabetes.”
III. Zepbound (Tirzepatide)
- Primary Role: Chronic Weight Management & Obstructive Sleep Apnea (OSA).
- The 2026 Edge: Zepbound is currently the “Weight Loss King” due to its dual-receptor activity. In December 2024, it also became the first medication FDA-approved to treat Moderate-to-Severe Sleep Apnea.
- Key Clinical Data (SURMOUNT-5): In the highly anticipated 2025 head-to-head trial against Wegovy, Zepbound proved superior, achieving a mean weight loss of 20.2% compared to Wegovy’s 13.7% over 72 weeks.
- Average Weight Loss: 20% – 22% at maximum tolerated doses.
4. The Rise of Oral GLP-1s: Rybelsus vs. The New Wegovy Pill

Historically, peptides like semaglutide were considered “un-pillable” because the stomach’s acidic environment and proteolytic enzymes (like pepsin) would destroy them before they reached the bloodstream.
The breakthrough that allows for both Rybelsus and the Wegovy Pill is SNAC technology (Sodium N-caprylate).
I. The Science: SNAC Technology
Both oral versions use SNAC as an absorption enhancer. SNAC works through a dual mechanism:
- pH Neutralization: It creates a localized “buffer zone” of higher pH around the tablet, protecting the semaglutide molecule from acid-catalyzed degradation.
- Transcellular Facilitation: It increases the lipophilicity of the gastric membrane, allowing the large semaglutide molecule to pass directly through the stomach lining into the blood.
II. Rybelsus (The Diabetes Forerunner)
- FDA Approval: 2019 (Type 2 Diabetes).
- Dosage: 3 mg, 7 mg, and 14 mg.
- Clinical Goal: Glycemic control (A1C reduction).
- Weight Loss Profile: While it does cause weight loss, it is modest (approx. 5% at the 14 mg dose).
- In the USA, it is often used “off-label” for weight loss, but it lacks the potency of its injectable siblings.
III. The Wegovy Pill (The 2026 Weight Loss Revolution)
- FDA Approval: December 22, 2025.
- Launch Date: January 2026 (USA).
- Dosage: 25 mg (and 50 mg in ongoing trials).
The Difference: The Wegovy Pill is the first oral GLP-1 specifically indicated for Chronic Weight Management.
The OASIS 4 Data: Clinical trials showed that the 25 mg daily pill achieves a mean weight loss of 16.6% in adherent patients-effectively matching the efficacy of the weekly Wegovy 2.4 mg injection.
| Feature | Rybelsus | Wegovy Pill (New) |
| Max Approved Dose | 14 mg | 25 mg |
| Primary Indication | Type 2 Diabetes | Obesity / Weight Loss |
| Average Weight Loss | ~4-5% | ~14-17% |
| Administration | Daily (Fasting) | Daily (Fasting) |
| Cardiovascular (MACE) | Approved for T2D | Approved for Obesity + Heart Disease |
IV. The “Pharmacist’s Warning”: The 30-Minute Rule
- The bioavailability of oral semaglutide is extremely low (less than 1%) compared to subcutaneous injection (89%).
- So, to ensure enough medication enters the blood and works to inhibit glucagon and slow the stomach emptying, patients must follow the strict administration protocol:
- Take on an empty stomach immediately upon waking in the morning.
- Use no more than 4 oz (120 mL) of plain water to swallow the pill.
- Wait at least 30 minutes before eating, drinking, or taking other medications.
5. Pharmacokinetics: Weekly (Injectables) vs. Daily (Pills).
The molecular structure of these agonists is engineered for longevity.
While natural GLP-1 has a half-life of roughly 2 minutes due to rapid degradation by the enzyme DPP-4, these synthetic versions are modified:
- Semaglutide (Ozempic/Wegovy): Features an albumin-binding side chain that shields it from enzymatic breakdown, extending the half-life to approximately 7 days.
- Tirzepatide (Zepbound): Utilizes a similar fatty-acid diacid side chain, resulting in a 5-day half-life, yet its steady-state concentration allows for effective weekly dosing.
The primary challenge with GLP-1 therapy is maintaining a “steady state” in the blood without overwhelming the gastrointestinal system.
I. Plasma Half-Life and Steady State
- Unlike older GLP-1s (such as Exenatide), which required twice-daily dosing, modern agents are engineered for long-term stability.
Semaglutide (Ozempic/Wegovy): Has a half-life of approximately 165–184 hours (about 7 days).
It takes roughly 4–5 weeks of consistent dosing to reach a “steady state” where the drug concentration remains constant in the blood.
Tirzepatide (Zepbound): Has a slightly shorter half-life of about 5 days.
This still supports weekly dosing but explains why missing a dose by more than 2-3 days can lead to a more noticeable “hunger rebound.”
II. Bioavailability: The 1% Hurdle
- Injectables: Subcutaneous bioavailability is approximately 89% for semaglutide and 80% for tirzepatide.
- Nearly all of the drug reaches the systemic circulation.
- Oral (The Wegovy Pill): Oral bioavailability is a mere 0.6% to 1%.
- This is why the 25 mg oral dose is required to achieve the same effect as the 2.4 mg injectable dose.
- If a patient drinks too much water or eats too soon, that 1% can drop to nearly 0%, rendering the pill ineffective.
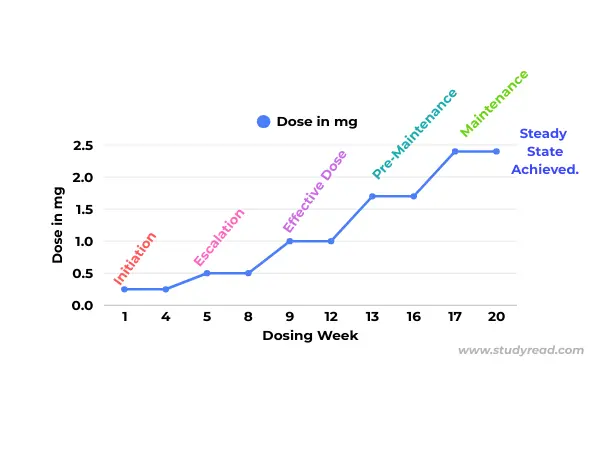
III. The Logic of Titration (The Escalation Schedule)
- The body’s GLP-1 receptors, particularly in the brain and gut, need time to “desensitize” to the sudden influx of hormones.
- Escalating the dose too quickly causes severe nausea and vomiting.
Injectable Titration (Standard 17-Week Path)
Most patients follow the “Rule of 4”—staying on a dose for 4 weeks before increasing.
- Month 1: 0.25 mg (Initiation – introduces the molecule).
- Month 2: 0.5 mg (Appetite suppression begins).
- Month 3: 1.0 mg (Weight loss accelerates).
- Month 4: 1.7 mg (Pre-maintenance).
- Month 5+: 2.4 mg (Full Therapeutic Maintenance).
Wegovy Pill Titration (New 2026 Protocol)
Because the pill is taken daily, the titration is measured in 30-day increments.
- Days 1–30: 1.5 mg (Starting dose).
- Days 31–60: 7 mg.
- Days 61–90: 14 mg.
- Maintenance: 25 mg (The 50 mg dose is currently reserved for “non-responders” in clinical settings).
IV. Clinical Note: Missing a Dose
- If < 5 days late: Administer the dose as soon as remembered.
- If > 5 days late: Skip the dose entirely. Do not double up.
- If >2 weeks have been missed: The patient’s GI tolerance has likely reset.
- They must call their provider and potentially restart at the 0.25 mg or 1.5 mg (oral) initiation dose to avoid severe “re-initiation sickness.”
6. Adverse Effects and Clinical Risks
- The pharmacological profile of GLP-1 agonists is generally well tolerated, but the rapid slowing of gut motility leads to a predictable “GI Burden.”
- As of 2026, clinical management focuses on distinguishing between transient discomfort and severe pathological risks.
I. Common Gastrointestinal (GI) Side Effects
These effects are dose-dependent and most common during the first 4–8 weeks of titration.
- Nausea & Vomiting (44%): The most frequent complaint.
- It is a direct result of delayed gastric emptying.
- Diarrhea (20%) & Constipation (24%): Caused by changes in intestinal transit time.
- “Sulfur Burps”: Delayed fermentation of protein in the stomach produces hydrogen sulfide gas, leading to “rotten egg” flavored burps.
- Dysgeusia: A metallic or altered sense of taste, affecting roughly 5-8% of users.
II. Serious Pharmacological Risks & “Black Box” Warnings
The FDA mandates strict warnings for specific high-risk categories.
- Thyroid C-Cell Tumors: A Black Box Warning exists for all GLP-1 and Dual-Agonists.
- Based on rodent studies, these drugs are strictly contraindicated for patients with a personal or family history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pancreatitis: Patients should be monitored for persistent, severe abdominal pain that radiates to the back.
- Acute Kidney Injury (AKI): Often a secondary complication of severe dehydration from vomiting and diarrhea.
- Maintaining a daily intake of $2-3\text{ liters}$ of water is a clinical priority.
- Gastroparesis (Stomach Paralysis): A rare but serious slowing of the stomach muscles.
- 2026 updates suggest this affects less than 1% of users but accounts for the majority of current litigation in the USA.
III. The 2026 “Quality of Life” Risks
- Sarcopenia (Muscle Loss): Without intervention, up to 40% of the weight lost can be lean muscle mass.
Precaution: Maintain a protein intake of 1.2g/kg of body weight and engage in resistance training.
- “Ozempic Face” & Hair Thinning: Rapid fat loss in the face can lead to a hollowed appearance, and the metabolic stress of rapid weight loss can trigger Telogen Effluvium (temporary hair shedding).
7. Clinical Management: Navigating the “GI Burden.”
- Because GLP-1s and Dual-Agonists work by slowing gastric motility.
- So, gastrointestinal (GI) side effects are not just “side effects”—but they are a direct extension of the drug’s mechanism of action.
I. Managing “Stomach Fullness” & Nausea
The Mechanism: By delaying the exit of food from the stomach (gastric emptying), these drugs can prolong the time food sits in the stomach by hours or even days.
- Mitigation Strategy: * Portion Control: Advise patients to stop eating before they feel full. Overeating while on a GLP-1 is the prominent cause of acute vomiting.
- The “Sulfur Burp” Fix: These occur when protein ferments in the stomach.
- Reducing intake of high-sulfur foods (onions, garlic, broccoli, eggs) and using digestive enzymes or gas-relief (simethicone) can provide significant relief.
- Bland Re-initiation: If a patient increases their dose (e.g., moving from 0.5 mg to 1.0 mg), a 48-hour “Bland Diet” (low fiber, low fat) can help the gut adapt to the new level of motility suppression.
II. The Sarcopenia Risk: A 2026 Clinical Priority
- A major concern in the USA throughout 2025 and 2026 has been Sarcopenic Obesity, i.e., losing significant weight but at the cost of vital muscle tissue.
The Problem: In rapid weight loss, the body often breaks down (catabolizes) muscle protein for energy. Without intervention, a 25% to 40% of the weight lost on Wegovy or Zepbound can be Lean Body Mass (LBM).
The 2026 Protein Protocol: * Target: Clinical guidelines now recommend 1.2-1.5g/kg body weight daily. For a 180 lb (82 kg) patient, this is approximately 100 to 125g of protein.
Resistance Training:
- Medication alone is no longer considered “Best Practice.”
- Providers are now prescribing at least 150 minutes of weekly activity, with a mandatory 2-3 sessions of resistance training to signal the body to preserve muscle.
Nutritional Monitoring:
- Recent 2026 cohort studies indicate that 12–15% of long-term GLP-1 users develop micronutrient deficiencies (specifically Vitamin D, B12, and Iron).
- Routine lab monitoring every 6 months is now the standard of care in high-end US obesity clinics.
8. Essential Precautions & Drug Interactions
| Category | Specific Precaution |
| Oral Contraceptives | Critical for Zepbound (Tirzepatide): Slowed absorption makes “the pill” less effective. Patients must use a backup barrier method for 4 weeks after starting and for 4 weeks after every dose increase. |
| Hypoglycemia | Increased risk when used alongside Sulfonylureas or Insulin. Dosages of these background meds often need to be reduced by 50% upon starting a GLP-1. |
| Gallbladder Disease | Rapid weight loss is a known trigger for Cholelithiasis (gallstones) and Cholecystitis. |
| Retinopathy | Rapid improvement in glucose can cause a temporary “paradoxical worsening” of diabetic retinopathy. |
| Pregnancy | Discontinue at least 2 months before attempting to conceive. Animal studies show potential for fetal skeletal and visceral anomalies. |
Conclusion: The Future of Weight Management
The 2026 landscape of GLP-1 agonists marks a pivotal shift from managing “blood sugar” to targeting the root hormonal drivers of obesity.
For healthcare providers and patients, the choice between Ozempic, Wegovy, Zepbound, and Rybelsus is no longer just about weight loss percentages-it is about choosing the right delivery system and metabolic pathway for the individual.
Key Takeaways for 2026:
- The Rise of Oral Potency: With the launch of the high-dose Wegovy Pill, the “bioavailability barrier” has been overcome, offering clinical results that finally rival those of weekly injections.
- Beyond the Scale: The success of a GLP-1 regimen is now measured by “Quality of Loss.” Preventing sarcopenia through high protein intake and resistance training has become as clinically important as the medication itself.
- The Dual-Agonist Lead: While semaglutide remains the gold standard for cardiovascular and renal protection, Tirzepatide (Zepbound) continues to lead in pure weight-loss efficacy due to its dual GLP-1 and GIP receptor activity.
As we look toward the next generation of “Triple-Agonists” and once-monthly dosing, the focus remains on safe titration and long-term adherence.
References:
- Semaglutide-NIH
- SELECT Trial (Wegovy Cardiovascular): Lincoff, A. M., et al. (2023). Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. New England Journal of Medicine.
- SURMOUNT-1 (Zepbound Weight Loss): Jastreboff, A. M., et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity.
- SURMOUNT-5 (Head-to-Head: Tirzepatide vs. Semaglutide): Eli Lilly and Company (2024/2025). Tirzepatide demonstrated superior weight loss compared to semaglutide 2.4 mg.
- OASIS 1 & 4 (The Wegovy Pill): Knop, F. K., et al. (2023). Oral semaglutide 50 mg once daily for adrenocortical weight management in adults with overweight or obesity. The Lancet.
- FLOW Trial (Ozempic & Kidney Disease): Perkovic, V., et al. (2024). Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. New England Journal of Medicine.
- FDA Approval of Wegovy for Heart Health
- FDA Approval of Zepbound for Sleep Apnea (2024/2025):
Medical Disclaimer: The information provided on Studyread.com, including text, graphics, images, and other material, is for educational and informational purposes only. It is not intended to be a substitute for professional medical advice, diagnosis, or treatment.
