Distillation is the process of separating a substance from a liquid mixture by evaporating and subsequent cooling into a separate chamber. There are different types of distillation, like
- Simple distillation
- Fractional distillation
- Steam distillation
- Vacuum distillation
- Azeotropic distillation
- Double distillation
- Triple distillation
Types of distillation in detail
Simple distillation
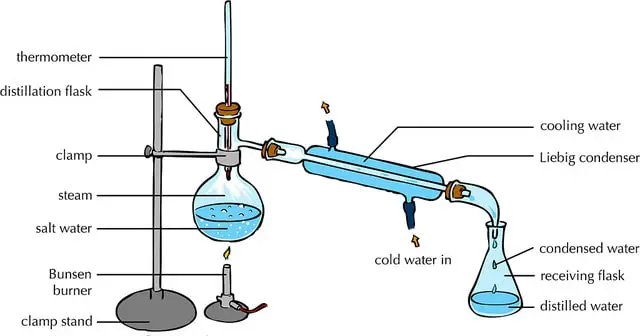
- Here, a liquid mixture is heated in a conical flask either with a burner or heating mantle.
- This heats up the liquid mixture, and the substance in the mixture with lower boiling points evaporates first.
- This vapor, rising from the top, is allowed to pass through a cooling chamber.
- The resultant liquid formed from cooled vapor is collected in a separate flask.
Fractional distillation

- This is similar to simple distillation but has an additional separation mechanism and a fractionating column in the equipment.
- As seen in the image above, a fractionating column is present between the flask and condenser.
- This fractionating column has beads inside the column, creating a hindrance to rising vapor.
- Due to this, some of the vapor gets in contact with the beads, converts back into liquid, and drains back into the flask with the liquid mixture.
- This process leads to the escape of only a vaporable portion of liquid into the condenser.
- Thus, this procedure helps obtain a pure form of substance that is not achieved by simple distillation.
Steam distillation
- As the name indicates, steam is used here for the separation of the liquid mixture.
- A steady flow of steam is passed into a liquid mixture in the round-bottom flask.

- This steam carries away one of the liquids from the container and gets cooled in the cooling chamber.
- The fluid formed by cooling the vapor is collected separately.
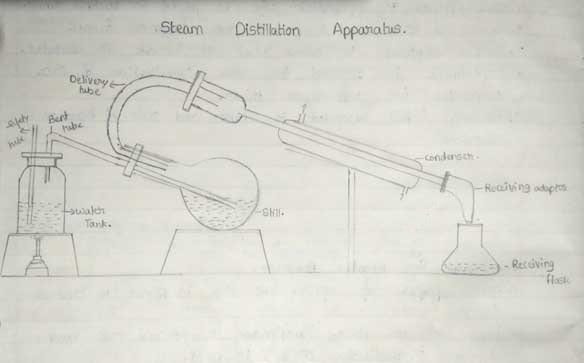
- This type of distillation is used to separate high-boiling substances from non-volatile impurities.
For example, turpentine oil boils in pure form at 160°C. When mixed with water, this turpentine oil boils at 96°C.
- At this temperature, the vapor pressure of water is 647mm Hg, and that of oil is 113 mm Hg.
- The combined sum is 760mmHg, which is equivalent to atmospheric pressure.
- So, to separate turpentine from impurities, it is mixed with water and then distilled by passing steam.
- The steam carries turpentine vapors and gets cooled in a condenser.
This is collected in a separate flask and can be easily separated since the water and turpentine oil are immiscible.
Vacuum distillation
- This is similar to simple distillation, with the only exception being the use of a vacuum to evaporate liquid at low temperatures.

- A liquid boils and converts into vapor when the vapor pressure is equal to the atmospheric pressure.
- In this procedure, low pressure or vacuum is applied to remove the atmospheric pressure.
- Due to this, evaporation occurs at relatively low temperatures. The vapor forms, which is then separated and cooled.
Thus, vacuum distillation is very useful for separating heat-sensitive substances.
Azeotropic distillation
- As the name indicates, this process involves specific changes in the material during the distillation process.
- When a mixture contains two liquids with similar boiling points, they cannot be efficiently separated using simple distillation.
- Hence, one of the substance’s boiling points is enhanced due to changes in its chemistry.
- Due to this, the other one boils first, and it is allowed to evaporate and cool into a separate flask.
- An example of azeotropic distillation is a mixture of water and ethanol. Both have similar boiling points.
- So, glycerin is added to this mixture to enhance the boiling point of water.
- Now, when this mixture is heated, ethyl alcohol boils first and evaporates out.
- The ethyl alcohol is collected separately as the vapors get cooled.
Double distillation
- Here, two simple distillation flasks are connected in a series, like in the image below.
- In this method, the condensate from the first distillation is allowed to reach the 2nd flask.
- In the 2nd flask, the condensate is again heated to form vapors
- This formed vapor again cools down to form liquid.
- This method is routinely used to obtain double-distilled water.
Triple distillation
- Here, the condensate from double distillation is heated to vapor to reach 3rd flask, as shown in the image below.

- The formed vapors are cooled like above to get a triple-distilled liquid. Triple distilled water can be obtained by this method.
- Triple-distilled water is widely used for research in analytical chemistry as it is free of toxins and other contaminants.
- This water is preferred for laboratory research like HPLC chromatography, making buffers, etc.
- This method of multiple distillation is said to remove dissolved salts and other substances, leading to the minimization of conductivity and the enhancement of the purity of water.
