Many compounds are identified and classified for proper use by chemists.
But due to many chemical experiments and space explorations, many new compounds emerge, which need to be identified.
Even if you find one, you can follow the few steps mentioned below to identify the unknown compound
Identifying Unknown Compounds
Compounds can be identified by two examinations, like
1. Physical evaluation
2. Chemical evaluation
Physical evaluation
This relies on physical appearance and characteristics like
- State
- Color
- Texture
- Smell (0d0r)
- Taste
- Feel
Physical properties like
- Crystalline or amorphous nature
- Solubility
- Melting point
- Boiling point
- Refractive index
Chemical evaluation based on
1. pH
2. Conductivity
3. Functional groups, etc.
By analytical techniques like
- Spectroscopy methods of analysis
- Chromatography method of analysis
Let’s see them in detail
State:
Materials are available in three states:
- solid,
- liquid, and
- gas.
So when an unknown substance is present, its state can give us an idea of what the compound could be.
Example:
Hard solids can be mostly metals,
while water and other solvents can be liquids.
Based on color:
- Most solids or solutions have some form of color.
- Liquids and gases are mostly colorless. But still, some possess color.
- The presence of color easily gives us an idea of what the compound is.

For example, black-colored solids indicate a carbon compound, such as coal, or a heavy metal, such as iron.
Based on texture and feel:
- Not all substances feel the same in touch.
- Some are smooth, rough, powdery, sticky, slimy, hairy, slippery, etc.
- Based on these observations, one can also identify the compound.
- A smooth substance can be a solid, semisolid, or even viscous liquid. Most metals are smooth, solid materials like aluminum, magnesium, copper, and gold.
- While oils and waxes are also smooth but hardened liquids.
- However, brittle solids may fracture under pressure. This could be wood, paper, glass, or other materials.
Based on odor or smell:
- Odor is one of the most powerful senses for identifying compounds.
- If a substance has no odor, it would be tough to rely on these methods.
- However, many compounds have odors and can be easily spotted and identified.
- A strong, solid metal can have a negligible odor. But powders, liquids, and gases can have some sort of odor.
Based on taste:
Our tongue can recognize four types of tastes:
- sweet,
- salt,
- bitter, and
- sour.
Compounds like sugars are mostly sweet, while unripe fruits like lemons and oranges can be sour or salty.
Some fruits, like bitter guard, can be bitter. Hence, taste helps to identify the compounds in most cases.
But it is not advisable to always do so, as it can lead to toxicity.
Physical properties:
This is characteristic of a particular compound and can be scientifically recorded.
These properties include
1. Crystal or amorphous nature:
- This is meant for solids and indicates their physical feel and appearance in the powdered form.

- Notice table salt, sugar, or even talcum powder.
- You will feel them as soft but hard particles that can be prickly; these are crystalline.
- When observed under a microscope, they will have a three-dimensional structure or lattice.
- The atoms and molecules would be arranged in a highly ordered microscopic structure.
- Their crystal properties can also be used to identify compounds using techniques such as X-ray crystallography.
- On the other hand, substances like camphor, charcoal powder are said to be amorphous.
- If you hold them in your hands, the compound would feel like a solid. But the powder is not hard and prickly.
- It can even be crushed into pulp and granules. The particles would not have a definite shape even when observed under a microscope.
- But they would have interconnected structural blocks.
2. Solubility:
- The unknown substance is dissolved in water. If it is not soluble, it is mostly a nonpolar compound.
- This can then be tested in other organic solvents, such as ethyl alcohol, acetone, and benzene.
- Compounds like carbohydrates, salts, acids, alkalies, and metal oxides are soluble in water. But polymers like plastics and resins are insoluble in water.
- This solubility can also be further evaluated as either completely soluble or partially soluble. But still, it helps to ascertain the polar nature of the substances.
3. Melting point:
- This test is specifically for solids.
- As you might have already known, a substance, when thoroughly heated, changes its state.
- A solid converts to a liquid and a liquid converts to a gas at a suitable temperature.
- The melting point is the temperature at which a solid converts to a liquid.
- This melting point is so specific to a compound as it depends on the internal arrangement of molecules and bonds.
- However, this melting point can vary slightly if the test substance is contaminated with impurities.
- This melting point can be ascertained by an oil bath and Kofler bench
Example:
The melting point of iron is 1811K, while that of Aluminum is 933.47K. The MP of paraffin wax is 310K.
4. Boiling point:
- Like melting points, most liquids have a specific boiling point. It is the temperature at which a liquid converts into a gas at the atmospheric pressure.
- Technically boiling point is always higher than the melting point.
Examples: The Boiling point of iron is 3134 K, of Aluminum is 2743 K.
Refractive index:
- The speed of light propagation through the material is called the refractive index.
- This is measured by hectometers.
- Mostly liquids, gases, and transparent solids like glass are estimated.
- This is a fundamental physical property of the substance. It is used to identify the substance, its purity, and also its concentration.
| Substance | Its refractive index |
|---|---|
| air | 1.000293 |
| acetone | 1.36 |
| benzene | 1.501 |
pH:
- If a substance is a liquid or gas, its pH can be estimated.
- Even a solid can be dissolved, and its pH can be noted.
- The pH indicates if a substance is acidic, basic, or neutral.
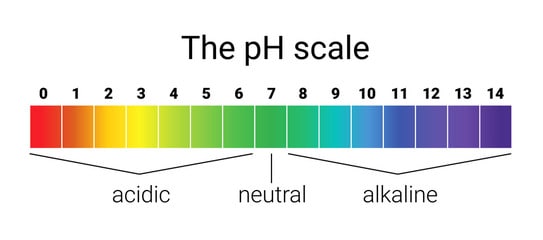
As you might have already known, acidic pH is less than 7, neutral pH is exactly 7, and alkaline pH is above 7.
Conductivity:
- It is the ability of a substance to conduct electricity.
- This is seen mostly in liquids, but even solids can be dissolved.
- At a given ionic strength, mass conductivity varies among substances.
Functional groups:
- This method is especially applicable for organic compounds.
- Functional groups are the parts of the molecule that determine the character of the whole substance.
These functional groups include
a) Hydroxyl groups
b) Ketone groups
c) Ester.
d) Ether, etc.
All these functional groups can be identified by specific chemical tests. Mentioning them is beyond the scope of this article.
Technical methods:
- These are highly specific and help to identify substances by comparison with a standard.
Spectroscopy methods of analysis:
- This is a method of analysis in which the behavior of a substance in the presence of light is estimated.
- It is estimated for its maximal absorbance, fluorescence, etc.
- Further, methods such as infrared spectroscopy can even determine the functional groups within the substance.
Chromatography method of analysis:
- This is basically a method of separation. After separation, the individual compounds are estimated by spectroscopy.
