Nuclear magnetic resonance spectroscopy is one that studies the spin changes at the nuclear level. This spin change occurs when a radio frequency energy is absorbed by the nucleus in the presence of a magnetic field.
Principle of NMR spectroscopy
In an atom with an odd mass number, the proton (nucleus) spins on its own axis. When an external magnetic field is applied, the spin shifts to precessional orbit with a precessional frequency. But still, the nuclei are in the ground state with its spin aligned with the externally applied magnetic field.
To this atom, if radio-frequency energy is applied such that the applied frequency is equal to precessional frequency, then the absorption of energy occurs leading to an NMR signal.
Since the energy is absorbed, the nucleus moves from the ground state to the excited state with its spin oriented in the opposite or anti-parallel direction.
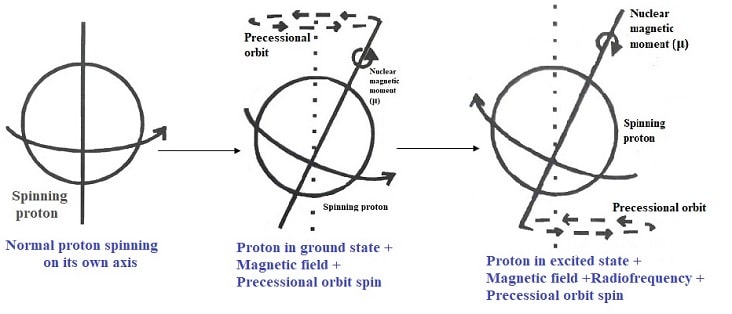
If the application of radio frequency energy is stopped, then the nucleus returns to the ground state with parallel orientation spinning is precessional orbit.
If even the magnetic field is removed, the nucleus will return to its normal spin on its own axis instead if precessional orbit.
Hence, application of magnetic filed only makes the nucleus to spin in the precessional orbit while the application of radio frequency energy leads to NMR signal.
This indicates that both the application of the magnetic field as well as the radio frequency energy are needed to produce NMR signal.
Instrumentation for NMR spectroscopy
An NMR spectrometer consists of 5 main parts like
1. Sample cell:
2. RF transmitter
3. RF detector
4. Sweep generator
5. Recorder.
Sample cell: This a test tube like glass apparatus placed in the sample cavity. It is of 25 cm long and 5mm in diameter. To provide uniform magnetic exposure to the sample, the cell is rotated at a speed of 30 revolutions per second.
RF transmitter: This is used to apply radio-frequency radiation to the sample. It can apply a frequency of 60, 90, 100, 220, 300 and 400MHz depending on the resolution of the instrument.
RF detector: It is meant to determine the unabsorbed radio-frequency radiation.
Sweep generator: This is not one which applies a magnetic field. But it is one which helps to modify the strength of the applied magnetic field.
Recorder: It helps to record the NMR signal received by the RF detector.
Magnetic field strengths of 14092, 21,120 and 23, 490 gausses are commonly employed.

Solvents:
Unlike other types of spectroscopy methods, here solvents need special attention.
The solvents must lack hydrogen atoms in their chemistry, should have magnetic isotropy (neutrality), be chemically inert, volatile to recover sample and inexpensive.
Since we are analyzing the organic compounds based on their hydrogen atoms (protons), the solvents with hydrogen can pose a problem. So, solvents which do not contain a proton are utilized for the NMR spectroscopy.
Examples of solvents used include Carbon tetrachloride (CCl4), Deuterated water (D2O), Deuterated methanol(CD3OD), Deuterated acetic acid (CD3COOD), Deuterated chloroform (CDCl3), etc. are used.
Reference standard: Since there are chances of many peaks in the NMR data. A reference peak is necessary for comparison and measurement. For this purpose, a substance called tetramethylsilane is used (TMS).
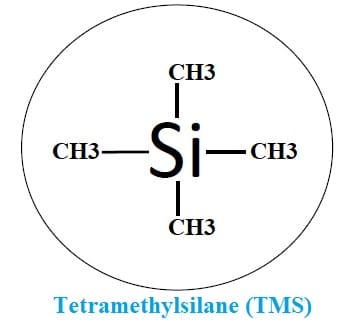
This TMS is bath chemically and magnetically neutral. It shows a single sharp and easily recognizable peak. It is soluble with most solvents and also volatile to recover the samples. Hence, it can also be used as an internal reference material.

Applications of NMR spectroscopy
1. Structural elucidation: Most organic compounds have hydrogen in them. NMR helps to identify the number of hydrogen atoms, their types, position and other characters. Thus it helps to understand the structure of a compound.
2. Quantitative assay of components: A component or more can be estimated without separation. Even the percentage of hydrogen in a compound, the chain length of polymers like polythene, moisture content, etc. can be analyzed.
3. Identification of properties of molecules like the isomerism, hydrogen bonding, tautomerism, etc. can be determined by using NMR spectroscopy
