Lab glassware is an essential part of experimental labs, as it is used in chemistry, biochemistry, biology, genetics, etc.
There are many types of labware made of different materials for use.
However, those made of glass are commonly used and suitable for all experiments.
Labware made of glass is preferred due to many advantages, like
- Chemical inertness to the sample and reaction mixture is taken.
- Ability to withstand high temperatures during heating, up to 500 degrees.
- Ease of cleaning, drying, and environmentally friendly disposal.
- It is low-cost and lasts longer.
- They can also be sterilized by heat if needed.
Due to the above reasons, lab glassware is quite famous. Let’s see them one by one.
Lab glassware names used in labs
- Glass Beaker
- Measuring cylinder
- Conical flask
- Test tube
- Round bottom flask
- Volumetric flask
- Filtration flask
- Funnel
- Separating funnel
- Burette
- Pipette
- Desiccator
Let us see their features in detail.
1. Glass beaker
- This is a plain and simple cylindrical container made of glass.
- It is transparent and has a flat bottom with a facility to pour liquid.
- You can find glass beakers in different sizes like 10ml, 25ml, 50ml, 100ml, 250ml, 500ml, and even 1000ml.
- The glass beaker has readings on the surface to indicate volume levels in the container.

This beaker finds its use in
a) To store liquids like solvents, solutions, reagent mixtures, oils, etc.
b) To mix a substance into liquid solvents.
c) To make solutions by rigorous stirring procedures
2. Measuring cylinder
- It is similar to a beaker but has a very small diameter and more height.
- It has graduations on the surface to indicate its liquid volume.
- It is widely used to take a desired volume of liquid into a beaker.

- Measuring cylinders find their use when the amount of sample to be measured is more than 5ml or 10ml.
If less than this, pipettes can be used for more accuracy.
Uses:
- To measure and take a desired volume of liquid.
- To make up the final volume of mixtures, small additions are made using a pipette.
3. Conical flask

- This is a conical-shaped glass apparatus with a round bottom.
- The conical flask does not contain graduated readings in most cases.
- It is mostly available in 25, 50, 100, and 250 ml sizes.
Uses:
- Widely used in titrations like acid-base, redox, complexometry, etc. Since the mixture requires constant stirring, the sample is taken in a conical flask, and the reactive agent is added from the burette drop by drop with a constant swirling of the flask and its contents till the endpoint.
- It can also be used for reactions involving heating. Since the mouth is narrow, the fumes of the reaction can be made to escape safely without exposing the lab interiors.
4. Test tubes

- These are the most commonly used glassware in the labs.
- Test tubes are mostly non-graduated, as one can add the desired volume from a pipette or burette.
- They are available in different sizes, like 5ml, 10mk, 15ml, 18ml etc.
- They have curved bottoms and are not flat like measuring cylinders.
- They are also required in large numbers as small amounts of reagents can be taken at a time.
- They need a stand to hold them in place while filled with liquids.
Uses:
- For storing small volumes of samples.
- For heating reactions, take a small quantity of mixture using a test tube holder.
- Most chemical tests are done using test tubes.
5. Round bottom flask

The round-shaped transparent flask has the ability to hold liquids and has a narrow mouth.
Round bottom flasks are available in shapes like 100ml, 250ml, 500ml, etc.
Uses:
- For the distillation of solutions, wherein the substance is taken in the flask and heated from the bottom.
- For mixing the substances in liquids.
6. Volumetric flask
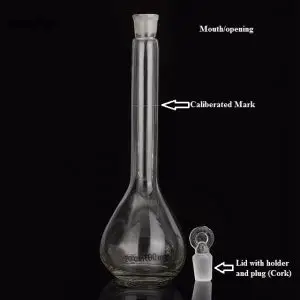
- It is a fixed-volume flask used to make molar solutions.
- The volumetric flasks are round at the bottom with a long, narrow neck.
- See the page for more details on the flask.
7. Filtration flask

- The flask is similar to a conical flask with a small vent on the side to connect with a vacuum.
Uses: This flask is especially needed for filtration and crystallization of extracts in the chemistry lab.
8. Funnel

- As we know, the funnel is a device to pour solvents and liquids into a narrow-mouth bottle.
- In the lab, one often needs transparent funnels to pour solvents, powders, and other liquids into other containers.
- These funnels are very useful as they minimize the chances of waste due to spillage.
Uses: This helps in the safe transfer of liquids and also prevents spillage and wastage.
9. Separating funnel
- Unlike other glassware, a separating funnel is a specially designed-apparatus for separating liquids.
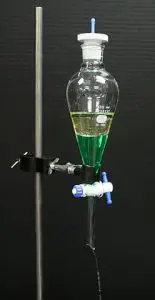
- A conical-shaped flask that has an inlet and outlet. It can hold liquid without leaks when closed with a stopper on top.
- When two or more immiscible solvents are mixed, the solvents get separated into two layers.
- The vent at the bottom of the flask can be opened, and individual solvents can be drained out.
Uses: This is useful for separating substances from a mixture based on their polarity or solubility.
Ex: Using petroleum ether, lipids can be separated from an aqueous extract.
10. Burette
- It is a long cylindrical-shaped glass tube with a stopper at one end.
- This burette has a uniform diameter along the length, with clearly marked graduation indicating volume.

- The stopper is fixed before the outlet.
- Through this, one can extract a desired volume of liquid from it.
- As shown in the image below, a burette needs a stand to hold it in place.
Uses: It is widely used in titrations. It is used to hold the titrant and add it to the reaction mixture (titrand) drop by drop.
11. Pipette

- Pipettes are glassware with a narrow path with graduations over the surface.
- They are used to transfer small amounts of liquids with precise volumes.
- The liquids are drawn into a pipette up to the desired volume and then transferred into another flask for further procedures.
- For safety, one needs to use pipette bulbs to suck the liquid instead of the mouth.
12. Desiccator:

- This is an enclosure used to store the chemicals without exposure to moisture.
- It is made of glass and has two chambers, one atop the other.
- The lower chamber mostly houses the desiccating chemicals meant for dehydration.
- Meanwhile, the upper chamber has a desiccating substance like anhydrous calcium chloride that can absorb moisture.
Frequently asked questions and answers.
When is the best time to clean glassware during the lab session?
I think it is best to clean glassware at the end of the lab session after all the experiments are over.
This way, they will be dry and clean to use in the next session.Why are most chemistry containers made of glass?
Glass is a hard and inert material that is least reactive to solids and liquids used in chemistry.
Since the chemicals we use, like acids, bases, oxidants, and halogens, are highly reactive. They can be stored well for a long time without being degraded.Why is acetone used to clean glassware?
Acetone is used to clean glassware for anhydrous experiments. I.e., for experiments that do not involve water.
As a solvent, acetone can clean and remove most chemicals, like water.
In most cases, the glassware is first cleaned with water, and then they are rinsed with acetone to remove the water stains.
🎓 Professor’s Study Tip: If you find memorizing these technical details difficult, I recommend this Top-Rated Medical & Science Study Aid. It uses AI to turn these notes into flashcards and practice quizzes automatically.

Thank you we love it
Ok will update..:-)
Catalogue number and price of each item would be more useful
Simple and precise explanation