Carbon belongs to the 2nd period of the periodic table. It is located in the ‘P’ block. Thus, the atom’s outermost electrons are present in the ‘P’ orbit. It can form bonds with itself which is termed as concatenation.
Electrons in carbon
Carbon is present in group 14 of the periodic table.
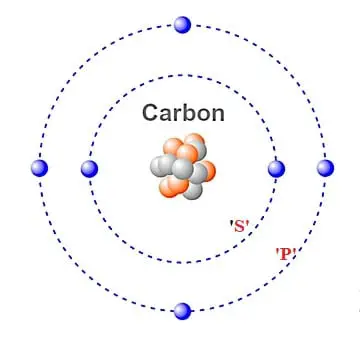
Carbon has an atomic number of six and a mass number of 12.01u. Thus, six electrons exist in a neutral atom of carbon. Two electrons are present in the inner orbit, and four electrons are present in the outermost orbit. The outermost orbital can hold a maximum of eight electrons. The electronic configuration of carbon is 1s2 2s2 2p2.
Thus, the ‘P’ orbit is the outermost orbit. Carbon needs to have a fulfilled p orbital to achieve stable gas configuration.
The closest noble gas neon (Ne) has the electronic configuration 1s2 2s2 2p6. Thus, the carbon needs to get 4 more electrons.
Carbon usually forms covalent bonds and shares electrons to achieve this stability. Carbon is neither an electropositive nor an electronegative element, so it forms covalent compounds.
Carbon has the unique property of concatenation, where it can form bonds with itself continuously in a chain or ring form.
Carbon dioxide (CO2)
Each carbon atom is bonded to two oxygen atoms. The carbon atom forms a double bond with each oxygen atom.
It shares 4 electrons with oxygen(2 from each) thus filling its p orbital.
One electron from the 2s orbital moves from 2s to 2p, forming 2 sp hybridized orbitals of carbon, which overlap with two p orbitals of the oxygen atoms to form 2 sigma bonds.
The two remaining p electrons form pi bonds.
Thus, a linear structure is formed. The central atom of carbon dioxide, which is carbon, is thus sp hybridized.
Ethene (C2H4)
Ethene contains two carbon atoms which are doubly bonded to each other. Each carbon atom is bonded to two hydrogen atoms each.
Each carbon thus shares two electrons with the carbon atom to which it is bonded.
The two hydrogen atoms bonded to each carbon atom share two electrons with the carbon atom (one electron from each hydrogen).
Thus, carbon shares four electrons and fulfills its p orbital. Each carbon atom is SP2 hybridized and has a triangular planar structure.
Methane (CH4)
Each carbon atom is bonded to four hydrogen atoms. Each carbon atom forms one covalent bond with each hydrogen atom.
It shares 4 electrons with hydrogen (1 from each), thus filling its ‘P’ orbit.
Here, the electronic configuration of carbon changes from 1s2 2s2 2px1 2py1 to 1s2 2s2 2px1 2py1 2pz1.
Thus, it can form 4 bonds and form a tetrahedral sp3 structure.
