Mass spectrometry is used to determine the mass and molecular structure of a compound.
Unlike other spectroscopy techniques, here the electromagnetic radiation is not used for the excitation of electrons. Further, there is also no phenomenon of electrons moving in between the ground state and excited state.
The principle of mass spectrometry is based on the ionization of sample molecules into a positive (+ve) state and separating them under the influence of electric and magnetic fields. This occurs in two steps as
- Ionization of molecules
- Separation of positively charged ions
Ionization of molecules: The sample molecules are bombarded with high-energy electron beams with 70ev. Due to this, an electron is knocked out from each molecule leading to the formation of +ve charged ions.
Separation of positively charged ions: The +ve charged fragments formed in the previous step are now subjected to positive potential leading repulsion. The +ve charged fragments get repelled and travel at high speed in a straight path.
Now either an electric field or magnetic field is applied so that the +ve charged molecular fragments travel in a curved path instead of a straight line.
However, when they take a curved path with high velocity, the fragments take different path lengths due to differences in radius of curvature. This difference in the radius of curvature is dependent on the mass size of the fragment.
This is given by the equations as
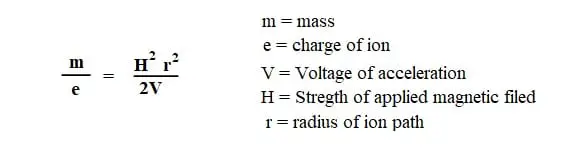
Since the values V, H are constant and e is equal to 1, which is a unit positive charge, the final effect would be like
the radius of curvature would be directly proportional to mass, i.e., m ∝ r.
Types of mass spectrometry
There are six different types like
- Electron impact mass spectrometer (EIMS)
- Chemical ionization mass spectrometer
- Filed ionization mass spectrometer
- Field desorption technique
- Plasma desorption technique
- Laser Mass spectrometer
Instrumentation of mass spectroscopy
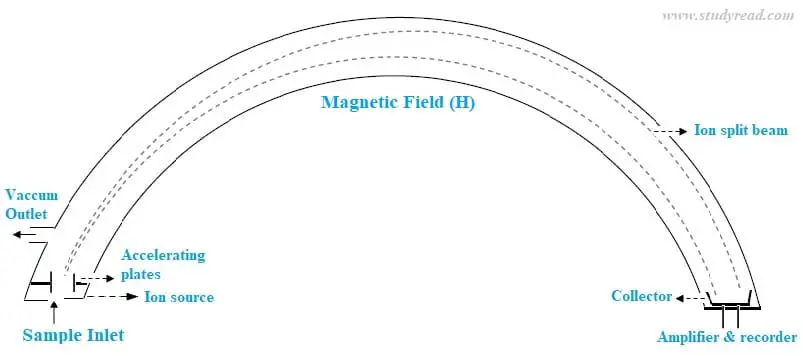
The mass spectrometer consists of a curved or horseshoe-shaped glass tube that evacuated. There is a sample inlet, electron bombarding source, and acceleration plate on one end. A sample collector is present at the other end of the glass tube. At the curvature magnetic field or electric field can be applied. A vacuum connect is also present to remove any air inside the glass tube.

Types of peaks in mass spectra
On analyzing any sample, few peaks are formed in the mass spectra. These peaks are of different types like
- Mass peak or parent peak or molecular ion peak is one with the highest m/e value on X-axis.
- The base peak is the one with the highest intensity on Y-axis.
- M+1 Peak these peaks are given due to ions like C13, H2, N15, S33
- M+2 peaks are those given by ions of O18, S 34, Cl37, Br 81, etc.

Application of mass spectroscopy:
Determination of compounds: Mass spectrometry helps determine the molecular weight, molecular formula and elemental composition of compounds.
Structural elucidation: Every molecule has a specific structure. This structure can be ascertained by this method. This is done by using the nitrogen rule.
To distinguish between Cis and Trans isomers of drug molecules.
Detection of impurities: Impurities can be determined by extra peaks obtained in the mass spectrum.
Analysis of proteins
Assessment of fragmentation patterns
Metabolism studies: When a drug is metabolized, its metabolic fragments can be from the blood, or a urine sample can be used to analyze its structure.
Forensic and toxicological applications
In case of death by poisoning, there are chances that only small quantities of toxin are present. Such small samples can be analyzed by mass spectrometry to identify the actual poison.
Gas chromatography-mass spectroscopy: Mass spectrometry is combined with gas chromatography to analyze explosives, environmental analysis, drug testing, etc.
