Aluminum is a bright lustrous metal widely obtained from nature.
In large quantities, it is isolated from the bauxite ore.
It is one of the highly available elements making up to 8% of the earth’s crust.
Aluminum’s atomic number is 13, while its standard atomic weight is 26.9815.
It has 13 electrons in the shells as 2, 8, and 3.
It is widely used by man in everyday life. This is because of aluminum’s characters like low density and high corrosion resistance.
It is well tolerated by animals and plants and hence it is less toxic.
Further, it can be easily recycled and disposal is environmentally friendly.
Aluminum properties
Aluminum properties are so distinct that, it is being widely used in industry, household, medicine etc.
Physical properties
1. Appearance: It is silvery, shiny and white in color. It is smooth and soft to touch. This property gives it steel like appearance, and the products made are attractive.
2. Weight and density: It is very light in weight. Its density is 2.7 g/cm3 due which it sinks in the water yet it is very light.
3. Conductivity: Aluminum has very good conductivity for both electricity and also heat. However, it is less conductive by at least 40% to Copper. But, it is highly used in place of copper due to the cost and weight.
4. Ductility: Unlike iron metal, aluminum can be easily molded. It does not break, crack or shatter when made into different shapes.

This feature makes it useful as foils, metal sheets etc.
5. Melting point: When heated, aluminum starts to melt at a temperature of 660.3°C or 1220.5°F
6. Boiling point: This melted solution when further heated, it starts to boil at 2470°C, 4478°F.
7. Magnetic properties: It is nonmagnetic by nature. But it is classified under paramagnetic substances.
Chemical properties of aluminum:
1. Alkaline nature: Like all the metals aluminum has alkaline properties.
Its oxides form alkalis like aluminum hydroxide.
However, these aluminum oxides react with both acids and alkalis. Hence they are called amphoteric oxides.
It reacts with acid to form salt aluminum

The oxide form reacts with the alkali to form
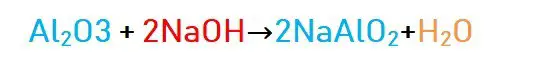
2. Reaction with water: Aluminum does not react with hot or cold water. But it reacts with steam to form aluminum hydroxide and hydrogen gas (g). 
3. With oxygen and air: Aluminum burns in oxygen to form aluminum trioxide (Al2O3).
4. Corrosion: Aluminum is covered with a coat of oxide over its surface. When the fresh surface is exposed to air, this coating is formed by a process called as passivation. Hence, it does not react with air.
5. Reducing properties: Aluminum undergoes displacement reaction. By this, it shows reducing properties. As you know, the reducing agent is one which gives up electrons.
When manganese dioxide is heated along aluminum powder this reduction is seen. The reaction leads to the formation of manganese and aluminum dioxide.

what are the physical properties of aluminum?