Radioimmune assay (RIA) is an immunological assay to analyze any antigen or antibody in the patient’s serum to diagnose the disease.
This is one of the most sensitive & specific methods of immune assays available.
It involves the competitive binding of radio-labeled antigen and unlabeled antigen to a high-affinity antibody.
The sensitivity range is 0.0006–0.006 µg antibody/ml.
The technique was developed by S. A. Berson and Rosalyn Yalow, and Rosalyn R. Yalow received the Nobel Prize in 1977.
It involves three principles that make it more specific & sensitive than other immune assays.
Immune Reaction
When a foreign biological substance enters the body’s bloodstream through a non-oral route, the body recognizes the specific chemistry on the surface of the foreign material as an antigen.
It then produces specific antibodies against the antigen so as to nullify the effects and keep the body safe.
The body’s immune system produces antibodies, so it is an immune reaction. Here, the antibodies or antigens bind and move due to chemical influence.
This is different from the Principle of electrophoresis, where proteins are separated due to charge.
A competitive binding or competitive displacement reaction
This is a phenomenon wherein, when two antigens can bind to the same antibody, the antigen with higher concentration binds extensively with the limited antibody, displacing others.
So here in the experiment, the radiolabelled antigen is allowed to bind to a high-affinity antibody.
Then, when the patient serum is added, unlabelled antigens start binding to the antibody, displacing the labeled antigen.
Measurement of radio emission
Once the incubation is over, then washings are done to remove any unbound antigens.
Then, the radio emission of the antigen-antibody complex is taken, and the gamma rays from the radiolabeled antigen are measured.
The approach to RIA
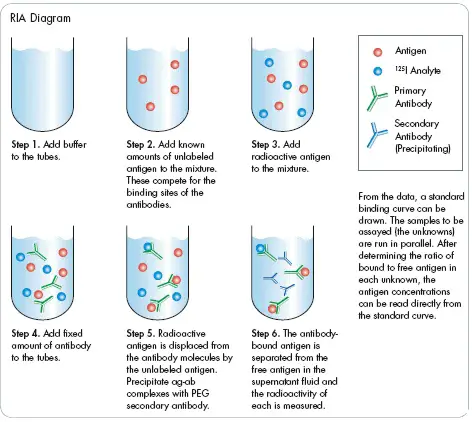
The first step to setting up an RIA is determining the amount of antibody needed to bind 50–70% of a fixed quantity of radioactive antigen (Ag*) in the assay mixture.
This antibody ratio to Ag* is chosen to ensure that the number of epitopes presented by the labeled antigen always exceeds the total number of antibody binding sites.
Consequently, unlabeled antigen (from patient serum) added to the sample mixture will compete with a radiolabeled antigen to bind to the limited number of antibodies.
Even a small amount of unlabeled antigen added to the assay mixture of labeled antigen and antibody will cause a decrease in the amount of radioactive antigen bound, and this decrease will be proportional to the amount of unlabeled antigen added.
To determine the amount of labeled bound antigen, the Ag-Ab complex is precipitated to separate it from free antigen (antigen not bound to Ab).
The radioactivity in the precipitate is measured.
A standard curve can be generated using unlabeled antigen samples of known concentration (in place of the test sample).
From this plot, the amount of antigen in the test mixture may be precisely determined.
Procedure of RIA
- The labeled antigen is mixed with an antibody at a concentration that saturates the antigen-binding sites of the antibody.
- Then, test samples of an unlabeled antigen of unknown concentration are added in progressively more substantial amounts.
- The antibody does not distinguish labeled from the unlabeled antigen, so the two kinds of antigen compete for available binding sites on the antibody.
- As the concentration of unlabeled antigen increases, more labeled antigen will be displaced from the binding sites.
- The decrease in the amount of radiolabeled antigen bound to the specific antibody in the presence of the test sample is measured to determine the amount of antigen present in the test sample.
- The antigen is generally labeled with a gamma-emitting isotope such as I125, but beta-emitting isotopes such as tritium (3H) are also routinely used as labels.
- The radiolabeled antigen is part of the assay mixture; the test sample may be a complex mixture, such as serum or other body fluids, that contains the unlabeled antigen.
The procedure requires small amounts of samples and can be conducted in small 96-well microtiter plates; hence, this procedure is suitable to determine the concentration of a particular antigen in large samples.
For example, a microtiter RIA can be used to screen for the presence of the hepatitis B virus.
RIA screening of donor blood has sharply reduced the incidence of hepatitis B infections in recipients of blood transfusions.
Radioimmunoassay is not widely used as of ELISA tests in health care.
Because RIA is a time-consuming method and also very expensive.

HOW CAN I CITE THIS ARTICLE?
Perfect explanation. Easy to understand
Wonderful concept clarity about RIA.
Never had been so simpler. Thanks
thank you. it helped me.
I have been helped to understand this(RIA) better. Thank you.
Simplified one… Better to refer
Thank you so much
comprehensive and straightforward
Thanks a lot its a good explanation and easy to understand
Thank you
excellent explanation….
very helpfull thnks
Easy to understand. Very helpful.Thanks
nice and easy explanation
excellent
Could you please explain the direct as well as the indirect RIA methods in detail as i could not find it anywhere else in detail
So good
Nice explanation
Thanks
you hvae coppyed same thing from kube .
Very nice easy to understand
good