Organic compounds are those compounds that have the element carbon in them.
Their chemistry and reactions are unique from other chemicals.
Some of them are naturally available, while others are synthesized by man.
Unlike inorganic compounds, there are thousands of organic compounds in the current day.
Due to the vastness of compounds, peculiar chemistry, and properties, they are studied as a separate branch, i.e., organic chemistry.
These compounds are of different types with different natures and phases.
Also, some of them exist as biomolecules in living beings, while others are available in nature.
Organic compounds contain mostly carbon, hydrogen, and oxygen.
Please refer to more details on organic chemistry for a better idea.
Examples of organic compounds
Solids: Diamond, coal, graphite, acids like (acetic acid, acetic acid), sugars, fats, etc.
Liquids: Ex: Benzene, pyridine, ethanol, acetylene, etc.
Volatile substances: Naphthalene (shows sublimation).
Gases: Methane, Acetylene, etc. See types of gases for more.
However, the best way to study organic compound examples is with the functional group in mind.
Functional groups are the key structures within a molecule that define the compound’s chemical properties.
These are the points of a molecule that undergo reactions and also contribute to physical properties.
The physical nature means the odor, state, and reactivity. So, based on the functional groups, we have different examples as
| Sl.No | Compounds | Functional group | Formula | Uses |
| 1 | Aliphatic compounds | CH | CH3-(CH2)n-CH3. | Ethylene makes polythene covers. Acetylene is used as a gas for welding. |
| 2 | Alkenes | CH=CH | H2C=CH2 | Gases, solvents, plastics |
| 3 | Alkynes | HC≡CH | HC≡CH | Gases. |
| 4 | Alicyclic compounds | -CH- | CH2-(CH2)n-CH2. | Di-ethyl ether is used as an anesthetic. |
| 5 | Carboxylic acid | -COOH | R-COOH | Help to form esters. |
| 6 | Ethers | R-O-R | CH3-(CH2)n-O-(CH2)-CH3. | Formaldehyde is used as a disinfectant to preserve biological samples, etc. |
| 7 | Esters | R-COO-R | CH3-(CH2)-COO-(CH2)-CH3. | All the cooking oils and lipids come under this category. |
| 8 | Aldehyde | R-CHO | CH3-(CH2)n-CHO. | For alcoholic drinks, as solvents, fuel, etc. |
| 9 | Ketones | R-C=O-R | CH3-(CH2)n-C=O-CH3. | Acetone is used to remove nail polish. It is also used as a solvent in chromatography. |
| 10 | Alcohols | R-CH2-OH | CH3-(CH2)n-COH. | For alcoholic drinks, as solvents, fuel, etc. |
| 11 | Amides | R-CONH2 | CH3-(CH2)n-CONH2. | They are part of proteins and are also used in many reactions. |
| 12 | Amines | R-CNH2 | CH3-(CH2)n-CHNH2 | As bases in acid-base titrations, as dyes, as drugs like chlorpheniramine (anti-allergy) |
| 13 | Amino acids | HOOC-CHR-NH2 | HOOC-CH2-NH2 | They are building blocks of proteins. |
| 14 | Nitriles | -CN | -C≡N | Nitrile rubber (latex-free gloves, automotive seals), acrylic fibers, and plastics |
| 15 | Sterols (Multi-cyclic structures with functional groups | 4-Fused Rings | There are many compounds used as solvents, medicines, catalysts, etc. | |
| 16 | Aromatic compounds | Planar unsaturated ring structures. | C6H5– | For alcoholic drinks, as solvents, fuel, etc. |
| 17 | Organic Halides | CH-X | Solvents, pesticides. |
Let us look at examples of organic compounds in detail.
Aliphatic compounds
- These are compounds that contain carbon and hydrogen.
- The bonds between two carbons can vary from one to three.
- These compounds can be enormous, like Hexane, a six-carbon chain {CH3-CH2-CH2-CH2-CH2-CH3}, heptane {CH3-CH2-CH2-CH2-CH2-CH2-CH3}, octane, an eight-membered carbon chain, etc.
Ex: Ethane {H3C-CH3}, Ethene {H2C=CH2}, Acetylene {HC≡CH}.
- These compounds are used widely.
Examples
- Ethene is used in making plastic bags, i.e., polyethylene covers.
- Acetylene gas is used in gas welding for joining metal parts.
Alicyclic Compounds
- As the name indicates, these compounds are similar to the above but form a ring in their structures.
- They are formed by a single bond between two carbon atoms in the chain.
- They are named Cyclopentane for a five-member ring, cyclohexane for a six-member ring, etc.
Compounds with functional groups.
- The same aliphatic compounds can have oxygen, nitrogen, sulfur, etc., as part of their chemistry.
- These chemical points in the molecule are called functional groups.
- These functional groups impart a distinctive character to the plain aliphatic chain or rings.
Functional groups have carbon, hydrogen, and oxygen.
Carboxylic acids
- This -COOH Bulky group ionizes, releasing H+ ions, indicating the molecule shows acidic character.
- The general formula of the carboxylic acid is R-COOH, and it ionizes to R-COO–.
- Carboxylic acid molecules are polar in nature due to the presence of two electronegative oxygen atoms.
- They also participate in hydrogen bonding due to the presence of the carbonyl (C=O) and hydroxyl groups.
- When placed in nonpolar solvents, these compounds form dimers due to hydrogen bonding.
- This increases melting and boiling points and changes other colligative properties.
- Unlike inorganic acids, which are liquids, these organic acids are solids.
- They are also not as strong as inorganic acids.
A few examples of carboxylic acids include
- formic acid (found in ants),
- acetic acid (found in vinegar),
- ascorbic acid (vitamin C), and
- citric acid (found in lemons).
- Tartaric acid
- Perchloric acid
Aldehydes
- These have an OH group linked to a carbon atom in the chain. Examples include formaldehyde and acetaldehyde.
- Formaldehyde is used to store biological specimens.
- They are also found in carbohydrate monomers.
Ketones
- These are structures in which an oxygen atom is linked to a carbon atom by a double bond, i.e., {C=O} in the molecule.
- Some examples include acetone, glucose, sucrose, fructose, etc.
- Acetone is used as a solvent.
- While Fructose and other sugars are used as food sources for carbohydrates.
Alcohols
- Alcohols are those molecules having the -OH moiety linked to carbon atoms directly.
- There are many types of alcohols based on molecular size. They are used as solvents due to their high polarity.
- However, not all of them can be used due to volatility issues.
- Ethyl alcohol,
- methyl alcohol, and
- propyl alcohol
are widely used due to their volatility and solubility properties.
- Also, ethanol is widely used for alcoholic beverages and even as a disinfectant to kill microbes.
Esters
- These are the molecules that form oils and fats. Examples include Arachis oil, sesame oil, mustard oil, etc.
- They have long chemical structures and are susceptible to oxidation when kept open to the air for long periods.
- Some of them are used as cooking oil, for massage, etc.
Ethers
- They are compounds with a profuse odor. Hence, they are named ethers.
- They have an oxygen atom linked to two carbon atoms. Ex: Diethyl ether (used as an anesthetic).
Fatty acids
- When esters break down, they release fatty acids and alcohols.
- They have -COOH structure in their molecules.
Ex: lauric acid, arachidonic acid, etc. These fatty acids are used to make soaps.
Amides
- These are the compounds formed by the reaction of acids and amines.
- Amides form substances like proteins, silk, and even drugs like paracetamol.
Amines
- These are basic and contain an ammonia moiety.
Examples: codeine is used for cough treatment.
- They are used as dyes to impart color to drugs and as indicators in titrations, etc.
Ex: sunset yellow, methyl orange.
Amino acids
- These structures have both carboxylic and amine moieties.
- There are many amino acids in the body. They help maintain the body by forming proteins.
Nitriles:
- These are organic compounds cyano functional group (−𝐶≡𝑁).
- They are highly polar and are mostly liquids or solids.
Check out Organic chemistry for dummies.
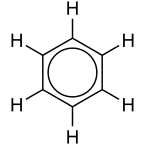
Aromatic compounds
- These compounds are cyclic but are unsaturated.
- They have an odor of their own. Ex: Benzene used as a solvent.
Steroid structures.
- These structures are quite complex, as seen in the diagram above.
- They form cholesterol and other structures.
- They are derived from fats and lipids. They are used as body booster drugs. Ex: betamethasone.
Alkyl halides:
- These are the ones that have halogens in their chemistry.
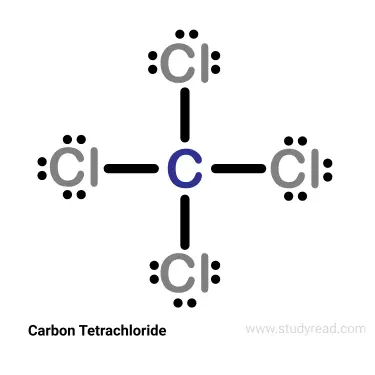
Examples: Carbon tetrachloride (CCl4).
Functional groups of organic compounds can be determined by specific chemical tests.
These tests help to know the nature of the unknown compound in the lab.
Also, read Best Way to Study Organic Chemistry.




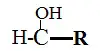


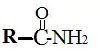
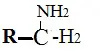
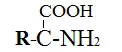


I enjoyed this lesson very much and need more information on this topic.
You Beautifully described about organic compounds
Minerals is an inorganic body found in the earth;ore_pertaining to minerals.
Its has also benifits,the miners collecting minerals,to get an ore.
Minerals is a substance which can be found on earth.