Chromatography is a separation technique used to determine the components of a mixture.
The Different Types Of Chromatography include
- Column chromatography
- HPLC (High-performance liquid chromatography)
- Gas chromatography
- Paper chromatography
- Thin-layer chromatography
- Ion exchange chromatography
- Size exclusion chromatography
- Supercritical fluid chromatography
- High-performance thin-layer chromatography (HPTLC)
- Liquid chromatography-Mass spectroscopy
- Gas chromatography – Mass spectroscopy
- HPLC – atomic absorption spectroscopy
- Gas chromatography – atomic absorption spectroscopy
Due to the variety of samples and their requirements, basic column chromatography was widely modified, leading to the development of different types.
Column chromatography
Initially, chromatography was developed as column chromatography.
It is the most widely used analytical technique in research labs and industries.
As the name indicates, there is a long column that is suspended in the air with the help of a stand.

The basic principle is that, into the column, a solid slurry (silica gel) is packed as a stationary phase, and the mobile phase liquid (organic solvent) is allowed to flow through.
This mobile phase takes the sample through the solid column. The components of the sample get separated due to relative affinity to the stationary phase.
It is quite easy to operate and a less expensive technique.
However, its applications are limited due to low efficiency.
High-performance liquid chromatography (HPLC)
This is a modification of column chromatography wherein a high pressure is employed for the effective separation of compounds.
Additionally, the stationary phase used is made up of small size particles to increase the surface area for effective separation. So, it is called a high performance instead of high-pressure liquid chromatography.
The principle is similar to column chromatography. The components of a mixture in the mobile phase travel in the column at different speeds. This rate of travel depends on the relative affinity of the components towards the mobile phase and stationary phase.
The mobile phase is pumped into the column at a defined pressure along with the sample. The use of either UV-visible spectroscopic detector potentiometric or any other suitable detector detects the sample components coming out of the column.
The application of pressure reduces the time of the run in the column. For better separation, enhanced stationary phases and mobile phases are incorporated.
Due to this modification, large molecules like proteins, fats, and small molecules like monoamines can be separated efficiently.
Those components with relatively similar chemistry, like monoamines, steroids, etc., can be analyzed well by HPLC.
It is very versatile and is used in forensic, drug, food, clinical, environmental, and biotechnological applications.
See the HPLC principle and method for more details.
Gas chromatography (GC)
In this type, a gas is used as a mobile phase, and a liquid layer coated on a solid support is used as the stationary phase. The volatile components of a mixture, based on their relative affinities to the mobile phase, are separated.
This technique is not widely used due to the limited number of stationary phases and expensive instrumentation and operation procedures.
It is quite useful for the analysis of a few drugs.
For details, check the gas chromatography principle.
Paper chromatography
As the name indicates, here, a specially designed filter paper is used as a stationary phase. The cellulose layers in the filter paper contain moisture, which acts as a stationary phase. Buffers and other solvents are used as mobile phases.
The principle of separation is a partition, but even an adsorption method can also be performed.
A very wide range of samples can be analyzed by using paper chromatography.
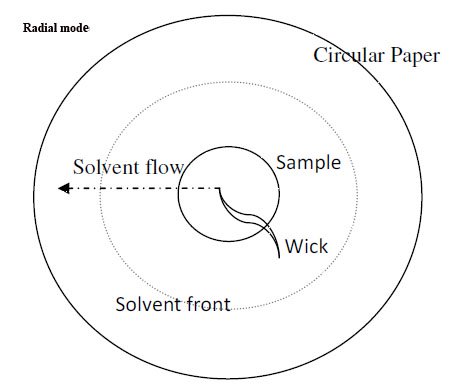
The sample dissolved in the mobile phase is marked on Whatman’s paper with a capillary tube or micropipette. Then, the paper is placed in the solvent vessel to allow it to percolate through the paper.
In doing so, it separates the sample over the paper surface, which is then subjected to color formation for identification.
Samples like carbohydrates, proteins, amino acids, vitamins, drugs, glycosides, alkaloids, and metabolites in blood and urine can be analyzed.
Thin-layer chromatography (TLC)
This method was first used for the separation of plant extract on a 2mm layer of alumina loaded on a glass plate. The principle of separation is adsorption. One or more samples are spotted onto the adsorbent coated over a chromatographic plate.

This plate is partially dipped into a mobile phase, as shown in the picture above.
The solvent moves along the adsorbent. The sample components move along with the mobile phase with different rates based on their affinity to the adsorbent.
This leads to the separation of components, which are detected with visualizing agents.
This thin-layer chromatography, similar to paper chromatography, can be used to analyze any type of sample. But it is routinely used to analyze plant extracts, drugs, carbohydrates, proteins, antibiotics, vitamins, etc.
Ion-exchange chromatography
Here, the mixture of charged ions is separated using an ion exchange resin.
There occurs a reversible exchange of similar ions between those present in the mobile phase and the ion exchange resin.
This exchange is based on the relative affinities. For a sample to be separated by this method, it should have charged particles such as anions or cations. As seen in the equation below,
Resin-H+ + M+ (mobile solution) ————–> Resin-M+ + H+ (in mobile solution)
The mobile phase is charged, and sample molecules with similar charges present on the charged stationary phase (resin) get exchanged.
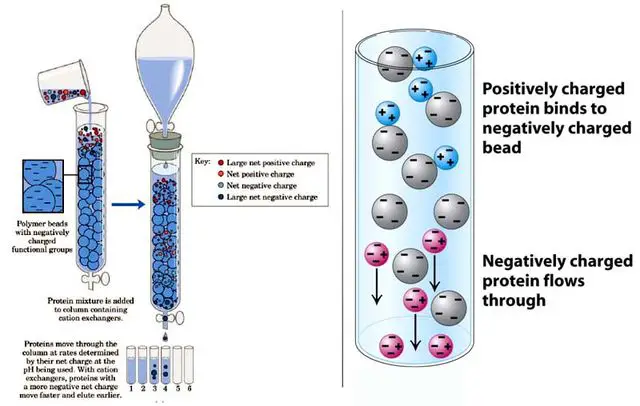
These ions are displaced from resin along with oppositely charged ions in the mobile phase and get eluted out of the column first. Then, a buffer solution is used to elute those molecules bound to the resin.
This ion-exchange chromatography is used for softening, deionization of water, purification of solutions, etc.
You can read more about ion exchange chromatography.
Size exclusion chromatography
Here, the column is loaded with charged gel having pores.
Sample particles, when poured along with the mobile phase, have to pass through the sieve-like network of the stationary phase.
In doing so, the larger particles elute out first and the smaller ones last.
The reason is the smaller ones take a longer path in the column stationary phase, while larger particles take a short path to elute out.
Find more about gel permeation chromatography.
Supercritical fluid chromatography
It is a normal phase chromatography with instrumentation similar to HPLC. Here, the mobile phase is mostly supercritical fluid-like carbon dioxide.
High-performance thin-layer chromatography (HPTLC)
As the name indicates, this is similar to TLC but more efficient.
Its efficiency is increased due to the use of small particles in the stationary phase, more choices of stationary phase material, auto-sampling technique, etc.
Read more about HPTLC.
LC-MS
This liquid chromatography equipment (HPLC), which is connected with Mass spectroscopy (MS) as a detector
GC-MS
Here, the Gas chromatography equipment column is connected with Mass spectroscopy as a detector.
HPLC-AAS
HPLC instrument column is coupled with an atomic absorption spectroscopy instrument.
Gas chromatography AAS
If the chemical sample is volatile and has metal ions to be analyzed, the sample is separated with gas chromatography and passed into atomic absorption spectroscopy.

I find it cool when you wrote a list of different chromatography techniques, especially about HPLC that features adding air pressure to separate proteins, fats, and small molecules efficiently. If I needed to use this method for my own sugar factory in the far future, I would buy my equipment, such as a headspace vial, from a local supplier. Doing this will help save time and energy to be used to produce more molasses and sugar.
this site is really great.i had to finish my project at the last moment and it had all the info i needed written in brief.this website is a life saver!!!
Short and precise.
Very very thank you for your usefull message for chromatography study
Thanks! I found it very nice! This is a good way of presenting. Can you send me the full material? G0 ahead!
Thanks for these article…
This article was very clear and easy for better understanding
This article is the best of them all
good article…plz send me a copy of this..so I can understand more..
Thank you for this such a good article.
I appreciate it.
kindly, send me a soft copy of this.
Nice combinations, your work has made my day lively.
Kindly send me a copy with the references.
Kept so simple and really interesting
this is very useful information
I would like to know who wrote this article when was it published for my lab report’s citation..
Thank you for this useful article.
Thank you for this such a good article.
I appreciate it.
kindly, send me a copy of this.
Thankyou once again.
thanks.
@Philip! Thanks for stopping by. Will try to add more shortly..
I really enjoy the explanation wish to know more about it
Thanks
i love this site
dr kumaraswamy! sent to ur mail
hi, good article. plese send me a copy of it.
regards.
dr kumaraswamy
thank you…for this….usefull..content….
thanks. This is very useful for my thesis.
sunita ranote! your compliment is inspiring…
artical in simple language.really nice n good work n moreover helpful n really thanks
Thanks and i find this article very useful.
Hi deepika soni! the matter is written based on personal collective idea on available types of chromatography. It is very easy for one to classify chromatography as he/she likes if they have overall idea of chromatography. By the way thanks for stopping by..
please send the reference of this matter
style of understanding is nice…………..
Muhammad Rizwan!glad that you found the article helpful..