Fluorimetry is a type of spectroscopy that measures the emitted radiation from a substance. This radiation is one that is emitted by the substance when the electrons transit from the excited state to the ground state.
The principle of fluorescence spectroscopy
In UV-Visible spectroscopy, the excitation wavelength is measured. But, here, the emission wavelength is measured.
When a substance is subjected to radiation of excitation wavelength, the electrons in the atom reach a singlet excited state. They then transit from singlet excited state to singlet ground state by emitting radiation with a specific wavelength. This emitted radiation is measured for analysis.
Instrumentation
The fluorescence spectroscopy instrumentation is similar to UV-Visible spectroscopy. It has the following
1. Light source: Xenon arc lamp, mercury vapor lamp and tungsten lamp
2. Monochromators: These help to separate light bands.
3. Sample cells: The sample cell is mostly quadrilateral. It is made of glass.
4. Detectors: This a photomultiplier tube that helps to detect radiation with low intensity.
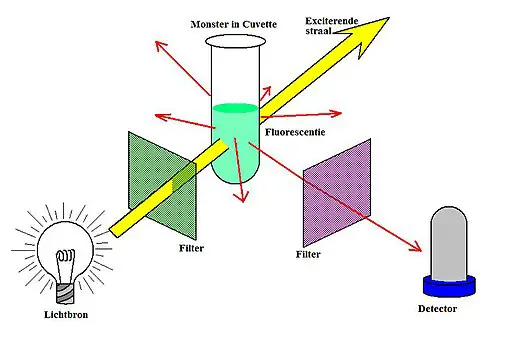
Fluorimetric instrumentation is of two types like filter fluorometer and spectrofluorometer.
Of them, the spectrofluorometer is advanced with high sensitivity and efficiency. But both of them have similar instrumentation as below.
Light source: The source should provide stable radiation with sufficient intensity over an entire range.
Xenon lamp: This is widely used in spectrofluorometers. Here the light is produced by passing electricity through ionized Xenon gas under high pressure. It provides a bright light with intense radiation over a wide range. Hence, most compounds can be analyzed.
Mercury vapor lamp: Here, mercury vapor is subjected to the high pressure of 8 atmospheres. It gives intense radiation over 350nm. At low pressures, this vapor also provides one more radiation at 254nm. But, this is preferred in inexpensive filter type fluorimeters.
Tungsten lamp: This lamp has radiation with low intensity. It is used when the excitation radiation is in the visible region.
Filters and monochromators: The light source produces polychromatic light. So, filters or monochromators are needed to convert them into monochromatic light. The filters have less resolution capacity and hence the light radiation has a bandpass of ±30nm. Consequently, they are inexpensive and used in filter fluorometers. In comparison, the monochromators have a very high-resolution capacity with a bandpass of just ±0.1nm. But, they are expensive and hence in spectrofluorometers.
Sample cells: The fluorometer cuvettes are small tube-like structures used to hold the sample for analysis. They are made of color corrected fused glass. All the sides of the cell have polished surfaces. The path length of the sample holder is 10mm.
Detectors: Photo-multiplier tubes are employed as detectors. These are efficient and accurate for radiations with low intensity.
Advantages of fluorescence spectroscopy
Sensitivity: Fluorimetry is a highly efficient spectroscopic with its ability to measure samples of small quantities. Samples concentrations as low as μg and ng/ml are determined.
Precision: The accuracy of the result is also high in this fluorimetry spectroscopy. A margin of up to 1% is achievable.
Specificity: UV Vis spectroscopy relies on just the excitation wavelength of the substance. In fluorescence spectroscopy, both excitation and emission wavelengths are characteristic. If two compounds could have the same excitation wavelength, they would differ in emission wavelength. Hence, the specificity for a compound is enhanced.
A wide range of compounds: This method is used to analyze many compounds. Even those compounds which do not have fluorescent property can be made by chemical conversion.
Disadvantages of Fluorimetry
Despite the many benefits of this method, some limitations exist. These are related to fluorescent intensity.
1. pH changes and the presence of oxygen can affect fluorescent intensity.
2. When a sample is subjected to UV absorption, it can undergo chemical changes due to the high energy of radiation.
3. Not all substances can be converted to be fluorescent. So, not all compounds can be analyzed by this method.
4. Presence of heavy metals and electronegative halides can change the fluorescent intensity.
Applications of fluorimetry
1. To analyze nucleic acids like DNA and proteins.
2. It is used to measure inorganic compounds containing metals or ions like beryllium, lithium, aluminum, zinc, etc.
2. It helps to analyze organic compounds like steroids, proteins, alkaloids, etc.
3. It is also a specified method to analyze medicines like morphine, quinine, indomethacin and vitamins like riboflavin, etc.
