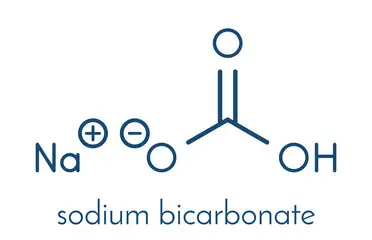
Sodium hydrogen carbonate is also known as sodium bicarbonate.
It has the chemical formula NaHCO3. It is a commonly used basic salt and is also known as baking soda.
It is a basic salt composed of a sodium cation (Na+) and a bicarbonate anion (HCO3−).
Sodium hydrogen carbonate is a white solid at room temperature. It is a crystalline compound, but it can also be found in the form of fine powder. Its other physical characteristics include the fact that it is odorless and soluble in water. Sodium bicarbonate has a slightly bitter taste owing to its alkaline nature. Sodium bicarbonate is found naturally occurring in the form of nahcolite. It is found as a precipitate in saline lakes and hot springs, etc.
When put in water, sodium bicarbonate dissociates into sodium ions (Na+), hydroxide ions (OH-), and carbonic acid H2CO3, which is a less strong acid. It ionizes weakly in water.
NaHCO3 + H2O → Na+ + OH– + H2CO3
It has a molar mass of 84. Sodium hydrogen carbonate is an amphoteric compound. It acts as both a weak acid and a weak base. However, the aqueous solutions of it are mildly alkaline due to the liberation of carbonic acid and hydroxide ions:
HCO3− + H2O → H2CO3 + OH−
Structure of sodium hydrogen carbonate
Each molecule of sodium bicarbonate consists of sodium cations and bicarbonate anions.

The bicarbonate ion consists of a central carbon atom, which is doubly bonded to an oxygen atom and singly bonded to a hydrogen atom. Carbon forms the fourth covalent bond with an oxygen anion, which is ionically bonded to a sodium cation.
Production of NaHCO3
- a) NaOH (diluted) + CO2 = NaHCO3
- b) 2 NaOH (conc.) + CO2 = Na2CO3 + H2O.
Na2CO3 + H2O + CO2 = 2 NaHCO3↓ (precipitates at 30-40° С).
c)NaCl + H2O + NH3 + CO2 = NaHCO3↓ + NH4Cl (Solvay process)
d)Sodium bicarbonate is produced on a large scale from sodium carbonate, which is available in nature.
Na2CO3 + CO2 + H2O → 2 NaHCO3
Reactions sodium hydrogen carbonate
2 NaHCO3 = Na2CO3 + CO2 + H2O (heat about 300 degree Celsius)
When put in water, it forms complexes and gives hydroxide ions
a)NaHCO3 (diluted) + 4H2O = [Na(H2O)4](+) + НСO3(-)
b)НСO3(-) + H2O ↔ H2CO3 + ОН(-).
Sodium bicarbonate is a basic salt and reacts with acids to give salt and release CO2 gas
a)NaHCO3 + НСl (aq) = NaCl + CO2 (↑) + H2O.
b)2NaHCO3(aq)+H2SO4(aq)→Na2SO4(aq)+2CO2(↑)+2H2O(l)
Above 50 °C (122 °F), sodium hydrogen carbonate decomposes into sodium carbonate, water, and carbon dioxide.
2 NaHCO3 → Na2CO3 + H2O + CO2 (↑)
Sodium bicarbonate reacts with acidic gases
a)6 NaHCO3 (conc.) + 3Cl2 = NaClO3 + 5 NaCl + 6CO2(↑) + 3H2O (boiling).
b)NaHCO3 + SO2 (↑) = NaHSO3 + CO2(↑)
Sodium bicarbonate reacts with acetic acid-producing sodium acetate, water, and carbon dioxide.
NaHCO3 + CH3COOH → CH3COONa + H2O + CO2(g)
Sodium bicarbonate reacts with bases like NaOH to produce carbonates
NaHCO3 + NaOH (conc.) = Na2CO3 + H2O
Other
4 NaHCO3 + 2CuSO4 = Cu2CO3(ОН)2 ↓ + 2 Na2SO4 + 3 CO2 ↑ + H2O (boiling).
Uses of sodium hydrogen carbonate
1. Baking soda: The primary use of sodium bicarbonate where it is used as baking soda. It is combined with cream of tartar(tartaric acid) to give baking powder which is used in doughs to make cakes, bread, etc. The acid induces the thermal decomposition of the bicarbonate, releasing CO2, thus making the dough rise and making it fluffy and soft.

The tartaric acid also has an additional use as the acid used in baking powder prevents a metallic taste from the sodium carbonate which is created in the chemical reaction.
2. Antacid in medicine: Another important use of sodium bicarbonate is its use as an antacid. It provides a basic medium in the stomach and neutralizes the excess hydrochloric acid. It may be used to treat the symptoms of stomach or duodenal ulcers.
3. To relieve constipation: A mixture of sodium bicarbonate taken with polyethylene glycol dissolved in water is an effective laxative used before gastric and intestinal surgeries.
4. In labs: NaHCO3, due to its mildly alkaline nature, is routinely used to neutralize acid spills in chemical laboratories
Sodium bicarbonate has varied purposes in cooking. It is used in breading food before frying to give the fried item a crispier flavor. Baking soda is used to soften beans for cooking. It is also used in some areas of the world to make the meat softer.
5. As disinfectant: Baking soda is a weak disinfectant and an effective fungicide.
NaHCO3 is used to raise the pH in pools and gardens. It is also used to modulate pH in blood when there are insufficient bicarbonate ions.
6. Fire extinguisher: Sodium bicarbonate can be used quite effectively in fire extinguishers in case of small electrical or grease fires. It acts as a dry chemical agent by releasing CO2, thus suppressing the fire.
7. Oral hygiene: Sodium bicarbonate is a popular chemical used for oral hygiene. It is used in toothpaste to whiten the teeth by removing plaque. It is also used in mouthwashes due to its abrasive and antiseptic properties.
8. Acidic drug poisoning: Sodium bicarbonate is used in case of toxicity due to acidic drugs like salicylates. In alkaline pH, the salicylates exist as ions that are water-soluble and get excreted in the urine. Thus, the administration of sodium hydrogen carbonate infusion helps to expel acidic drugs faster. Thus, it is used in the medical field to treat aspirin overdose, acidosis, hyperkalemia, etc. It is also used in combination with local anesthesia.

Hi ken, I suppose it would be just carbonated water. It might just have carbon dioxide in water.
I recently read that an old woman survived the 1918 flu pandemic. She and her family were the only survivors in a s village. She revealed that her whole family regularly took soda. I assume that she meant soda water, which contains Sodium hydrogen carbonate. Is this correct?