Alcohol in general means ethyl alcohol with a chemical formula of C2H5OH.
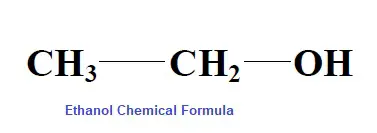
Ethanol is widely used as a part of hot drinks and beverages by man.
It is obtained by fermentation of sugar molasses on a large scale.
When consumed, it reaches the brain and creates a sense of well being and excitement.
See the article on the positive and negative effects of alcohol.
So, people consume ethanol regularly, which could lead to health problems.
However, besides drinking, there are many uses of alcohol in medicine, industry, research, etc.
Uses of alcohol in medicine
Alcohol is being used in medicine for ages. It has powerful antimicrobial, astringent, sedative, and rubefacient properties.
Treatment of methanol poisoning
When excess methanol is consumed, the person will have terrific symptoms leading to death. The methanol in the body converts to formaldehyde and formic acids, which causes pain and irritation all over. The sting of ants causes intense pain as it has formic acid. Imagine the extent of pain when formic acid is formed in the body due to methanol poisoning.
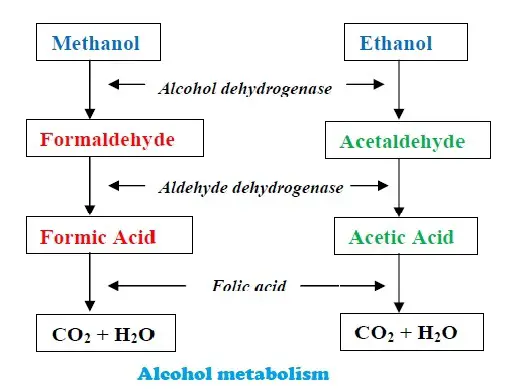
In this emergency, giving ethanol acts as an immediate antidote.
As seen in the picture above, Methanol is broken down into formaldehyde and then formic acid. Both of them are toxic to the body. But when ethanol is given, the rate of methanol is reduced. Ethanol has a higher affinity for the enzyme aldehyde dehydrogenase. So, it competes for the enzyme in place of methanol. Decreased formation of formaldehyde reduces the toxic effects on the body, sustains life, and gives more time to remove it from the body.
Other anti-dote like Fompezole can be used as it does the same function. But ethanol is inexpensive, easily available, and also more compatible in the body.
As rubefacient
70% ethanol and even Isopropanol have rubefacient character. When applied to the skin, it leads to the dilation of blood vessels and then redness. In this condition, the applied skin area is tender and has greater absorption property. This can be used to enhance drug absorption from ointment applied to the skin. Further, this relieves acute pain locally where applied.
To reduce pain
In case of severe pain like cancer, alcohol is injected near the nerve endings to destroy the nerve. This leads to a lack of pain from that nerve.
As Antiseptic
When an injection is to be made, a cotton swab soaked in alcohol is used to clean the surface. Then after injection, it is left there to see that there is no bleeding. This is done to clear the area of injection from any bacteria or microbes. Further, this cleaning removes any dirt in the site of injection.
Further, any surgical blades, needles, forceps can be cleaned with ethanol to remove microbes from them.
Alcohol at 70% is antiseptic and it can kill the microbes by denaturation of their surface proteins.

Diluent
Tinctures are alcoholic extracts of drugs. When a substance is in low concentration, it cannot be given as such for the person. Hence, to enhance the volume of the drug, alcohol is used as a diluent.
Since alcohol at such low doses is safe to the body and also sterile, it acts as a safe diluent. Especially in homeopathy, drugs are extracted with alcohol. Further, they are diluted with it for easy administration. Besides alcohol, lactose is also used as a diluent. However, alcoholic extract tinctures are also administered to patients.
Preservative
The tinctures, which are alcoholic extracts of plant and animal material, have a long half-life. This is because alcohol is a good preservative.
Uses of alcohol in industry
1. As a solvent
Ethanol, like water, is a good solvent for many polar compounds. This helps the solid material to be concentrated into a solution of the desired concentration. This solution is used for further processing like filtration, distillation, or removing impurities.
2. For extraction
Alcohol is a good polar solvent used to extract active ingredients from any raw material like the plant or other solids. This extraction is usually done by use of steam distillation connected to a soxhlet extractor.
Due to the low boiling point, alcohol easily evaporates and then percolates again through the solid material. In this way, there can be effective extraction.
3. Cleaning
Alcohol is one of the best solvents used for cleaning in the industry. Its properties of antimicrobial effects and solubility are effectively used.
4. Sterilization
Alcohol is one of the good chemical sterilizing agents used for sterilization.
5. De-staining
When stains catch any cloth material in the industry, alcohol is used to remove stains.
Uses in Biological studies
For Staining
In-plant tissue and animal tissue staining alcohol are used frequently.
Sanitation
Since biological substances are prone to microbial contamination, the use of alcohol clears them of microbes.
Ethanol uses in chemistry.
Alcohol is also widely used in chemistry for
1. Crystallization and extraction
A chemical synthesis involved a series of chemical reactions. The final product is in a liquid form. To extract it out as a solid, it is recrystallized and dried. This re-crystallization for some compound require ethanol.
2. Solvent for Analysis
Chemistry employs many analytical techniques like chromatography, HPLC analysis, etc. These analytical techniques require the separation of a component of the mixture. Ethanol, methanol, and isopropanol are widely used as different combinations of mobile phases.
Uses of alcohol in daily life:
Besides the above technical uses, alcohol is also widely used in daily life.
1. Clean marker stains
We tend to use markers often. The use of alcohol can easily remove these marker stains.
2. As automobile fuel

Ethanol is mixed with petrol to minimize pollution, save foreign exchange, and also provide greater energy. (Many countries import fossil fuels spending their foreign reserves)
3. In lamps
Alcohol lamps are used in laboratories due to their non-suit flames.
4. Beverages
Alcohol is used for recreation in the form of alcoholic beverages. It is a type of social lubricant.
5. Alcoholic inks
These inks have a pigment or dye dissolved in alcohol. When these inks are put on a tile, metal, or other surfaces, the mark is left. Alcohol dries quickly, leaving the pigment behind. These alcohol inks are used in making the background of stamping and cards.
6. Nail polish remover
Nail polish is easily removed by ethyl acetate or acetone. But if it is not available or its odor is irritating, then try ethanol.
It takes a longer time to remove, but it definitely removes the alcohol. Though I have never seen it, some claim to do so.
7. Rocket Propellant
Previously, ethyl alcohol was used as a combination of rocket fuels. It was used along with liquid oxygen diluted with water. Due to the availability of new and more efficient fuels, it is no longer used.
So there are many uses of alcohol, and this list may not be exhaustive.
