Visible spectroscopy is also called as colorimetry. This means it measures colored light (color+metry= color+ measurement).
The Principle of Visible spectroscopy
This is a study of the absorption of visible radiation within the wavelength range of 400nm to 800nm by any colored substance.
Every colored substance absorbs light radiation of different wavelengths. When we plot a graph of the wavelength of radiation vs. absorbance, a curve is derived which shows the wavelength at which maximum absorption takes place. This is termed as “λmax” and is unique for every compound. This λmax is qualitative aspect and helps to identify the substances.
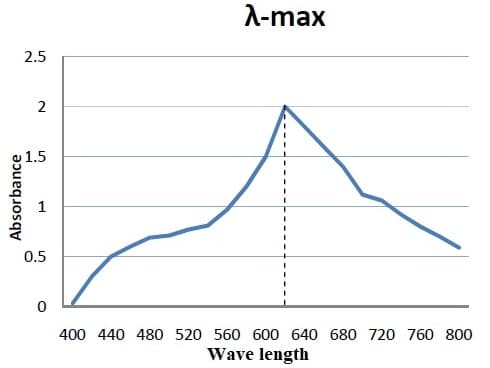
When the sample is exposed to the visible radiation, it absorbs light of a particular wavelength. If the concentration of the sample is high, the absorbance if high and vice versa.
So this is a type of spectroscopy is called as visible spectroscopy as relies on visible light for measurement.
This absorption is explained by two laws viz.
Beer’s law: The intensity of the beam of monochromatic (single colored) light decreases exponentially with an increase in the concentration of the absorbing substance arithmetically.
Lambert’s law states that the rate of decrease in intensity of monochromatic light with the thickness of the medium is directly proportional to the intensity of incident light.
Further details can be seen in the principle of spectroscopy.
Instrumentation of visible spectroscopy:
There are two types of instruments like simple colorimeters and sophisticated spectrophotometers. Their sensitivity and range differ, but the basic components for both remain the same.
1. Source of light: Any lamp which can produce a visible spectrum in the range of 400 to 800nm can be used. But it should have an adequate intensity and be free from fluctuations.
The common sources of light are tungsten lamp and carbon arc lamps.
a) Tungsten lamp: This lamp is widely used as is found in colorimeters and spectrophotometers. It consists of a tungsten filament in a vacuum bulb.
b) The carbon arc lamp: This is meant for high-intensity radiation. This consist of two carbon rods in free air. When the rods are touched to each other under low voltage, the lamp gets ignited. An electric arc if formed in between and they are slowly drawn apart.
Across the gap of rods, the electric current gets heated and maintains the arc. The carbon at the tip of rods vaporizes due to heat. This carbon vapor is luminous and produces a bright light.
The carbon arc lamp can also produce light over the entire range of the visible spectrum. But it is difficult to maintain and handle compared to the tungsten lamp.
2. Filters and monochromators: The visible white light has many wavelengths and colors. So filters and monochromators are used to filter unwanted wavelengths. The filters are of two types as
a) Absorption filters:
b) interference filters:
Absorption filters: These filters are less expensive and find a place in most of the colorimeters. They are made of either glass or dyed gelatin. They absorb unwanted radiation and transmit the remaining radiation.
These filters are easy to use and construct and also inexpensive.
The problem with them is less accuracy due to high bandpass of ± 30nm. For example, if we want a wavelength of 530nm, these filters pass radiation ranging between 500nm or 560nm.
Interference filters:
Here in between two parallel reflecting silver films, a dielectric spacer film is present. This spacer film is made of CaF2, MgF2 or SiO.
When light radiation is allowed to pass through the filters, the incoming radiation gets interfered by reflected radiation from the second film.

This constructive interference leads to the formation of monochromatic radiation. The wavelength of radiation obtained is given by the equation as below.

The order number mentioned in the equation indicates the thickness of the dielectric spacer films made of either CaF2, MgF2 or SiO.
These interference filters are efficient than the absorption filter as they have a lesser bandpass of ± 10nm only.
These filters are also inexpensive, and use of additional filters can cut of undesired wavelengths. But, the transmission and intensity of radiation could become low.
Monochromators
These are more sophisticated and accurate than the filters. They are of two types as
a) Prisms. (1. Refractive types 2. Reflective type)
b) gratings. (1. Diffraction 2. Transmission type)
They are expensive but very accurate. They have a bandpass of ± 0.1nm and have a higher range like 400 to 1000nm wavelength.
Sample cells: These are cuvettes which hold the sample under test. They are made of color corrected fused glass or polystyrene so that the light is not absorbed. Sample cells made of glass can be used for both aqueous and nonaqueous samples while those made of polystyrene cannot be used for organic solvents.
The sample cell should be completely transparent such that light falls and passes through it. These cells are mostly either cylindrical or rectangular. The internal path length is 0.1 cm or 1 cm or rarely even 10cm. They can hold 0.5ml to 5ml of liquid samples.
Detectors: Once the monochromatic lights pass through the sample cell, part of it is absorbed by the sample. The unabsorbed light is allowed to fall onto a detector for measurement. The detectors are of different types like
1. Photovoltaic cells
2. Phototubes
3. Photomultiplier tubes.
Of the above three, photomultiplier tubes are highly sensitive and accurate. So they are used in spectrophotometers while others are used in colorimeters.
When the photon from the unabsorbed radiation falls on to the detector, it is converted into an electrical signal and measured.
Types of instruments
The visible spectroscopy instruments are of two types as single beam and double beam types.
The single beam types are again as colorimeters and spectro-colorimeters. These single beam spectrophotometers have a single cell for sample alone.
While the double beam photometers have two sample cells, one cell is meant for the test sample and other is meant for reference solutions. This includes spectrophotometers with advanced filters, detectors and recorders.
Applications of visible spectroscopy:
Spectroscopy has many applications in fields like diagnosis, medicine manufacture, chemistry, research, etc.
1. For quality control of samples: In industry, many products are manufactured, and there are chances of contamination. Colorimetry can be used to assess the quality and purity of the finished product. The presence of impurities gives additional peaks other than λmax of the substance.
2. Quantitative analysis: Th percentage concentration of a substance in a given sample can be determined by absorbance characteristics
3. Structural elucidation: The structure of complex molecules like the organic compounds can be done by using colorimetry. The absorbance spectrum of the test substance can be compared with that of known ones.
This helps to define the most probable structure of the given test sample. But due to the availability of modern methods like mass spectrometry, I.R and NMR methods this method is not preferred.
4. Determination of dissociation constants. For substances like the buffers and acid-base indicators, the dissociation constant pKa can be determined.
5. Determination of elements, functional groups.
6. For monitoring of titrations. If either the titrant or the reacting test substance has absorbance properties, spectrophotometers can be used to monitor the end point of the titration.
