Chemistry is a significant branch of science that deals with matter and its properties.
The subject is quite vast and detailed for study. So it has been diversified into branches like
- Organic chemistry
- Inorganic chemistry
- Biochemistry
- Physical chemistry
- Medicinal chemistry
- Analytical chemistry
- Nuclear chemistry
- Environmental chemistry
- industrial chemistry
- Forensic Chemistry
- Synthetic chemistry
Since all the branches are helpful to man, they are also offered as part of different courses and degrees.
These branches of chemistry are helpful in fields like medicine, technology, food, and the environment. Thus, they are pretty essential to study.
What are the Branches of Chemistry
1. Organic chemistry
As the name implies, it deals with something related to carbon compounds.
There are millions of organic compounds in nature, so a separate branch is dedicated to their study.
Though it is true, in scientific terms, it is the study of carbon compounds. It includes the study of all the possible compounds which have carbon in them.
However, some define organic chemistry as the study of compounds having carbon, hydrogen, and oxygen.
Because pure carbon compounds like graphite, coal, and diamond are non-reactive, they are least studied in this branch of chemistry.
There is a vast number of compounds that have diverse chemical properties. They find their uses in medicine, food, agriculture, sterilization, disinfection, and also research.
Examples of compounds studied in this branch include; carbohydrates, benzene, amino acids, vinegar, phenol, etc.
For a complete list, read examples of organic compounds along with their structure.
What is studied: In general, the study includes synthesis, identification, purification, breakdown, and also methods to discover new compounds and their applications.
2. Inorganic chemistry
This is the chemistry that studies compounds devoid of carbon.
This branch of chemistry has a massive list of chemicals that are naturally found in nature. But some of them are also synthesized in the laboratory.
However, the number of compounds under this inorganic chemistry is lesser than those of organic chemistry.
The inorganic compounds find their use in medicine, food, agriculture, and also technology.
Examples of compounds in this branch include
Acids: Hydrochloric acid, sulfuric acid.
Salts: Sodium sulfate, potassium chloride;
Alkalies: Aluminum hydroxide, calcium hydroxide, etc.
Redox compounds: Potassium permanganate, potassium chlorate
3. Biochemistry
This chemistry, as the name implies, is the study of biological chemistry.
Biochemistry deals with chemistry happening inside the living bodies of animals and plants.
This subject is huge and plays an important role in medicine, agriculture, poultry, fisheries, etc.
It helped in understanding the actual physiology of our bodies and the cause of diseases.
It deals with four significant biochemicals like
- Carbohydrates (monomers)
- Proteins
- Fats (lipids) and
- Nucleic acids (DNA, RNA).
And also a few supporting elements like enzymes, vitamins, and hormones.
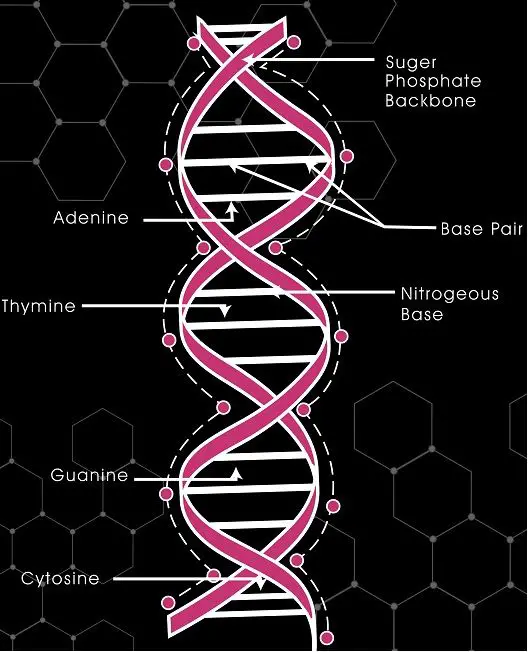
In plants, it explains the process of photosynthesis, respiration, transpiration.
While in animals, it describes their physiology and function. This includes processes like digestion, metabolism, excretion, respiration, and nerve conduction.
Its applications in daily life include glucose estimation (diabetic patients), fermentation, etc.
4. Physical chemistry
As the name indicates, it deals with the physical properties of chemicals or substances.
These include gaseous laws (Dalton law), thermal conduction in liquids, gases, solids. The conductivity of electrolytes (used in batteries), liquids, etc.
It also deals with sublimation, melting point, boiling point, the crystal structure of compounds, etc.
This chemistry finds its applications in other branches of chemistry besides daily applications in industries, automobiles, etc.
5. Medicinal chemistry
Chemistry plays a major role in medicine. Most medicines are of chemical nature. They are either organic or inorganic compounds.
They have therapeutic potential due to their chemical nature.
Hence this chemistry tries to study the therapeutic property in the compound, emphasizing its ability to bind to receptors, enzymes, bringing changes in the body’s chemistry, etc.
The study includes an evaluation of SAR (structural activity relationship).
This means a change in a functional group or the position of an atom or functional group in the structure leads to better efficiency and fewer side effects.
The study also includes the discovery of new compounds for medicine. Besides, new methods to synthesize the compound at less cost are explored.
6. Analytical chemistry
This chemistry deals explicitly with the analysis of chemicals. Analytical chemistry plays a vital role in the quality control of products in the industry.
Also, in research, analysis helps to identify and also explore the properties of compounds.
There are so two types of analysis the qualitative and quantitative analysis.

The qualitative analysis deals with the estimation of the quality of the compound and the determination of impurities.
While quantitative analysis estimates the amount of the main compound in the given sample.
Most methods of analysis estimate both qualitative and quantitative aspects. The study describes different methods of analysis like
- Titrimetric analysis by using titrations
- Chromatography
- Spectroscopy
- Gravimetry
- Electro-conductive methods
- Electrophoresis etc.
7. Nuclear chemistry
This branch of chemistry deals with radioactive elements. Their behavior like fission, fusion reactions, etc.
This chemistry finds application in making atomic bombs, atomic energy for generating electricity.
Also, see Branches of Physical Science.
8. Environmental chemistry
This involves studying the chemical, biochemical and basic mechanisms of pollutants in the environment and different ecosystems in nature.
It incorporates knowledge of different sciences like aquatic, soil, atmosphere, food, social, economic, biological, physical, and chemical correlations.
It aims to study methods to minimize pollution and its impact on nature.
9. Industrial chemistry
This deals with the transition of raw material to finished products of sufficient quantity using safe chemical processes.
The process can involve heating, mixing, dissolving, crystallization, filtration, evaporation, distillation, and other methods.
10. Forensic Chemistry
This is the study that involves the application of principles of chemistry in legal testing to identify unknown substances.
They aim to find evidence to help in testification.
They use different methods which are least destructive to the sample for the identification.
These methods involve gas chromatography, HPLC, spectrophotometry, volumetry, etc.
11. Synthetic chemistry
This aims to synthesize new materials by using different methods from existing materials.
It employs different chemical reactions, solvents, organic and inorganic compounds.
This chemistry finds huge application in the synthesis of medicines, organic compounds, and other essential chemicals.

Thanks and welcome for the knowledge encryption to me.
This really didn't help me out that much… I need the branch of science that chemistry and physics belongs to
It's such an interesting topic to learn. Thanks to you for adding to my knowledge.
thanks very much it has really helped me
HI kelly, thanks for stopping by and you are welcome again.
I am a 9th grade homeschool student and I was given the choice to write an essay about what I’d like to. I chose science cause I love it. I’ve always been interested in chemistry and chose to write about it. This article really helps me out! It was detailed enough to give me bases for my topic! Thank you so much!
It helps me a lot as a STEM students in shs. Thank you for this article I’ve learned a lot about the branches of chem.
It is such a very interesting way to get the best knowledge of the topics. Thank you for helping me..